Predict the products of each precipitation reaction. Balance the equation, and then write the net ionic equation.
Question:
Predict the products of each precipitation reaction. Balance the equation, and then write the net ionic equation.
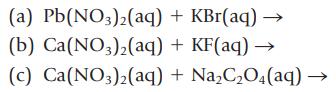
Transcribed Image Text:
(a) Pb(NO3)2(aq) (b) Ca(NO3)2(aq) (c) Ca(NO3)2(aq) + KBr(aq) → + KF(aq) → + Na₂C₂O4 (aq)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 44% (9 reviews)
To predict the products of each precipitation reaction youll need to identify whether an insoluble c...View the full answer

Answered By

Gabriela Rosalía Castro
I have worked with very different types of students, from little kids to bussines men and women. I have thaught at universities, schools, but mostly in private sessions for specialized purpuses. Sometimes I tutored kids that needed help with their classes at school, some others were high school or college students that needed to prepare for an exam to study abroud. Currently I'm teaching bussiness English for people in bussiness positions that want to improve their skills, and preparing and ex-student to pass a standarized test to study in the UK.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Predict the products of each precipitation reaction. Balance the equation, and then write the net ionic equation. (a) NiCl(aq) + (NH4)S(aq) (b) Mn(NO3)2(aq) + Na3PO4(aq)
-
Predict the products of each acid-base combination listed. Assume that a neutralization reaction occurs. a. HCl and KOH b. H2SO4 and KOH c. H3PO4 and Ni(OH)2
-
Predict the products of each acid-base combination listed. Assume that a neutralization reaction occurs. a. HBr and Fe(OH)3 b. HNO2 and Al(OH)3 c. HClO3 and Mg(OH)2
-
Ballard Ltd makes three products A, B and C. Each passes throughtwo departments: Machining and Assembly. Budgeted production ineach department by each productUnitsMachiningAssemblyPr 2 answers
-
The fundamental problem with any attempt to limit climate change is that the countries whose growth poses the greatest threat to the planet are also the countries that can least afford to pay the...
-
Dorothy Kelly sells life insurance for the Prudence Insurance Company. She sells insurance by making visits to her clients homes. Dorothy believes that the number of sales should depend, to some...
-
In a diesel engine, the fuel is ignited by (a) spark (b) injected fuel (c) ignitor (d) compressed air heat
-
The COSO internal control framework identifies five internal control components. Which of those components is most relevant to the procedures that SOX mandates public companies establish to enable,...
-
Encouraging team productivity can be a major issue in many organizations. Inclusive and Exclusive are the two common management styles most organizations exhibit. Discuss the effectiveness or...
-
Oxalic acid, H 2 C 2 O 4 , which is found in certain plants, can provide two hydronium ions in water. Write balanced equations (like those for sulfuric acid on page 142) to show how oxalic acid can...
-
Decide whether each of the following is watersoluble. If soluble, tell what ions are produced when the compound dissolves in water. (a) NiCl 2 (b) Cr(NO 3 ) 3 (c) Pb(NO 3 ) 2 (d) BaSO 4
-
The article "The Prediction of Corrosion by Statistical Analysis of Corrosion Profiles" (Corrosion Science, 1985: 305-315) suggests the following cdf for the depth X of the deepest pit in an...
-
Soccer Co . Soccer Co . has a group of companies operating within NSW . Payrolls for the month of August for each company are as follows: Soccer Importer $ 1 7 5 , 0 0 0 Soccer Holdings $ 1 1 8 , 0 0...
-
Farr Demolition stock sells for $23 per share. The firm has a rights offer outstanding for new equity shares. DeAndrea currently owns 200 shares. She just received one right for every share she owns....
-
On August 1 , 2 0 2 2 , Colombo Company s treasurer signed a note promising to pay $ 1 2 2 , 1 0 0 on December 3 1 , 2 0 2 2 . The proceeds of the note were $ 1 1 5 , 8 0 0 . Required: a . Calculate...
-
List 3 different types of market segmentations. Let's hypothesize that you are the CEO of a hockey equipment company. Who would likely be your main market segment? Explain your answer. List 3...
-
Using a discount rate of 4.5% compounded annually, a pension fund estimates that the present value of its assets and liabilities are $14 million and $13 million, respectively. The duration of the...
-
On January 1, 2011, Portico Company acquires 8,000 shares of Sauder Company by issuing 10,000 of its common stock shares with a par value of $10 per share and a fair value of $70 per share. The price...
-
Use critical values to test the null hypothesis H0: 1 2 = 20 versus the alternative hypothesis H0: 1 2 20 by setting a equal to .10, .05, .01, and .001. How much evidence is there that the...
-
Predict the major product(s) for each reaction below. (a) (b) (c) 1) LAH 2) , 1) PhMgBr 2) H,0
-
Starting with cyclopentanone and using any other reagents of your choosing, identify how you would prepare each of the following compounds: (a) (b) (c) (d)
-
Predict whether the following compound will be aromatic, nonaromatic, or antiaromatic. Explain your reasoning.
-
Coco Co. produces two products in the following two decentralised departments: The Processing Department: every 1 kg of raw cocoa beans is roasted and refined into cocoa solids without any weight...
-
Match each definition with its related term by selecting the appropriate term in the dropdown provided. 1. Actual Accounting System Term 2. Direct Labor Efficiency Variance 3. Direct Labor Rate...
-
What is the overall impact of adding the hammer to Tenalpina's product lines? How many hammers must be sold to "keep the company profits the same?"In other words, not decrease profits. Is purchasing...

Study smarter with the SolutionInn App


