Predict the products of each precipitation reaction. Balance the equation, and then write the net ionic equation.
Question:
Predict the products of each precipitation reaction. Balance the equation, and then write the net ionic equation.
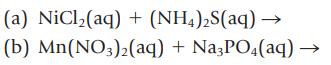
Transcribed Image Text:
(a) NiCl₂(aq) + (NH4)₂S(aq) → (b) Mn(NO3)2(aq) + Na3PO4(aq) →
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 40% (5 reviews)
Lets predict the products of each precipitation reaction write th...View the full answer

Answered By

Mugdha Sisodiya
My self Mugdha Sisodiya from Chhattisgarh India. I have completed my Bachelors degree in 2015 and My Master in Commerce degree in 2016. I am having expertise in Management, Cost and Finance Accounts. Further I have completed my Chartered Accountant and working as a Professional.
Since 2012 I am providing home tutions.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Predict the products of each precipitation reaction. Balance the equation, and then write the net ionic equation. (a) Pb(NO3)2(aq) (b) Ca(NO3)2(aq) (c) Ca(NO3)2(aq) + KBr(aq) + KF(aq) + NaCO4 (aq)
-
Predict the products of each acid-base combination listed. Assume that a neutralization reaction occurs. a. HCl and KOH b. H2SO4 and KOH c. H3PO4 and Ni(OH)2
-
Predict the products of each acid-base combination listed. Assume that a neutralization reaction occurs. a. HBr and Fe(OH)3 b. HNO2 and Al(OH)3 c. HClO3 and Mg(OH)2
-
. Current price of stock is $20 and expected price after one year is 22.5. If investor required return is 18%. What percentage of dividend should company pay?
-
France, in addition to its occasional stabs at strategic trade policy, pursues an active nationalist cultural policy that promotes French art, music, fashion, cuisine, and so on. This may be...
-
A sociologist studying New York City ethnic groups wants to determine if there is a difference in income for immigrants from four different countries during their first year in the city. She obtained...
-
Which of the following does not relate to spark ignition engine? (a) ignition coil (b) spark plug (c) distributor (d) fuel injector
-
Find the tension in the two cords shown in Fig. 9-45. Neglect the mass of the cords, and assume that the angle θ is 33o and the mass m is 170 kg.
-
In organizational behavior, understanding the factors that predict organizational performance is crucial for managers and researchers. Numerous theories and studies have attempted to shed light on...
-
Oxalic acid, H 2 C 2 O 4 , which is found in certain plants, can provide two hydronium ions in water. Write balanced equations (like those for sulfuric acid on page 142) to show how oxalic acid can...
-
Decide whether each of the following is watersoluble. If soluble, tell what ions are produced when the compound dissolves in water. (a) NiCl 2 (b) Cr(NO 3 ) 3 (c) Pb(NO 3 ) 2 (d) BaSO 4
-
The nominal interest rate associated with an ordinary general annuity is 7% compounded annually. Rounded to the nearest 0.001%, what is the corresponding periodic rate of interest that matches the...
-
Determine whether the ordered pair (8,5) is a solution to the following system. 5x-4y = 20 2x + 1 = 3y
-
Meyer Appliance Company makes cooling fans. The firm's income statement is as follows: Sales (7,000 fans at $20) $140,000 Less: Variable costs (7,000 fans at $8) 56,000 Fixed costs 44,000 Earnings...
-
Type of Bond Yield 1- year 0.2% 2- year 0.3 3- year 0.5 Using the expectations theory, compute the expected one-year interest rates in (a)the second year (Year 2 only) and (b)the third year (Year...
-
Fill in the missing values: Do not enter commas , % or $ signs. Remember to use 2 decimal places for all of your answers. Calculating Cost of Food Sold Food Sales $ 1 1 5 , 2 5 0 . 0 0 Opening...
-
The resolution of a telescope is ultimately limited by the diameter of its objective lens or mirror. A typical amateur astronomer's telescope may have a 6 . 0 5 in diameter mirror. What is the...
-
Whitney Company acquires an 80% interest in Masters Company common stock on January 1, 2011. Appraisals of Masters assets and liabilities are performed, and Whitney ends up paying an amount that is...
-
Cobb Manufacturing Company uses a process cost system and average costing. The following production data is for the month of June 2011. Production Costs Work in process, beginning of the month:...
-
Determine whether each of following ions is aromatic, nonaromatic, or antiaromatic. a. b. c. d. 0:
-
Explain the vast difference in pK a values for the following two apparently similar compounds. pk, = 16 pk, = 36
-
Predict which compound will react more readily in an S N 1 process, and explain your choice. Br -Br
-
Apex Limited (APL) is a management consultancy company headquartered in Singapore. After the successful implementation of a share option plan for the senior management team in 20x1, the company has...
-
7)A certain city covers an area of 62.294 mil 2 . How many square kilometers is this ?Use the conversion factor 1 mile = 1.61 km 8) A projectile is fired at 422.8 mi/hr. How fast is that in meters...
-
What is its unit price? Let's think of a batch of 100 baked goods...what is its total price? Speculate how much is the cost to bake (AND sell) that 1 batch (100 units)? Calculate its related gross...

Study smarter with the SolutionInn App


