Oxalic acid, H 2 C 2 O 4 , which is found in certain plants, can provide
Question:
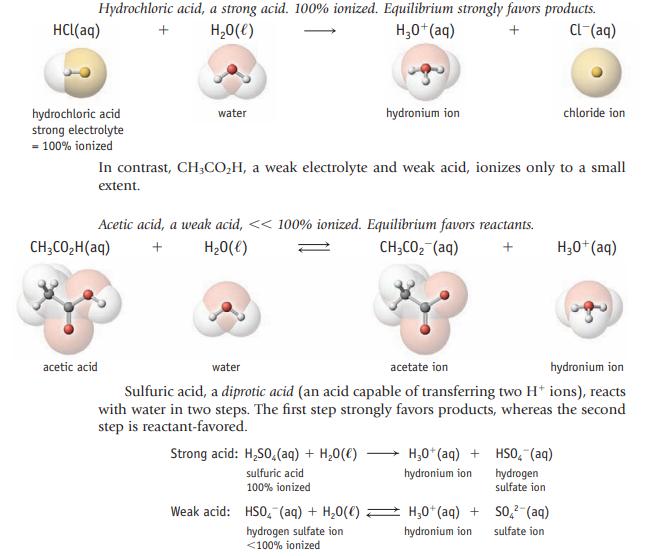
Oxalic acid, H2C2O4, which is found in certain plants, can provide two hydronium ions in water. Write balanced equations (like those for sulfuric acid on page 142) to show how oxalic acid can supply one and then a second H3O+ ion.
Data given in page 142
Transcribed Image Text:
Hydrochloric acid, a strong acid. 100% ionized. Equilibrium strongly favors products. + H₂O(l) H₂0+ (aq) + CL-(aq) HCl(aq) hydrochloric acid strong electrolyte = 100% ionized water CH3CO₂H(aq) acetic acid In contrast, CH3CO₂H, a weak electrolyte and weak acid, ionizes only to a small extent. Acetic acid, a weak acid, << 100% ionized. Equilibrium favors reactants. + H₂O(l) CH3CO₂ (aq) + hydronium ion Strong acid: H₂SO4 (aq) + H₂O(l) sulfuric acid 100% ionized Weak acid: HSO (aq) + H₂O(C) hydrogen sulfate ion <100% ionized water acetate ion hydronium ion Sulfuric acid, a diprotic acid (an acid capable of transferring two H+ ions), reacts with water in two steps. The first step strongly favors products, whereas the second step is reactant-favored. H₂0+ (aq) + hydronium ion chloride ion HSO₂ (aq) hydrogen sulfate ion H₂0+ (aq) + SO² (aq) hydronium ion sulfate ion H3O+(aq)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
Oxalic acid H2C2O4 is a weak diprotic acid meaning it can donate two hydronium ions H3O in water The dissociation of oxalic acid into its two hydronium ions can be represented by the following balanced equations First dissociation of oxalic acid H2C2O4aq H2Ol H3Oaq HC2O4aq Second dissociation of oxalic acid HC2O4aq H2Ol H3Oaq C2O42aq In these equations H2C2O4 represents oxalic acid H3O represents the hydronium ion ...View the full answer

Answered By

Emel Khan
I have the ability to effectively communicate and demonstrate concepts to students. Through my practical application of the subject required, I am able to provide real-world examples and clarify complex ideas. This helps students to better understand and retain the information, leading to improved performance and confidence in their abilities. Additionally, my hands-on approach allows for interactive lessons and personalized instruction, catering to the individual needs and learning styles of each student.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Phosphoric acid can supply one, two, or three H 3 O+ ions in aqueous solution. Write balanced equations (like those for sulfuric acid on page 142) to show this successive loss of hydrogen ions. Data...
-
Petitioner South Florida Water Management District operates a pumping facility that transfers water from a canal into a reservoir a short distance away. Respondents Miccosukee Tribe of Indians and...
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
Please help Gary earned \( \$ 97,000 \) as an executive. Gary, who is single, supported his half sister, who lives in a nursing home. His half sister had no income during the year. Gary received the...
-
Many countries have value-added taxes-taxes that are paid by producers, but are intended to fall on consumers. (Theyre basically just an indirect way of imposing sales taxes.) Such value-added taxes...
-
All Greens is a franchise store that sells house plants and lawn and garden supplies. Although All Greens is a franchise, each store is owned and managed by private individuals. Some friends have...
-
Carburettor is used for (a) S.I. engine (b) C.I. engine (c) Gas engine (d) none of the above
-
Winter Corporation has just completed its comparative statements for the year ended December 31, 2012. At this point, certain analytical and interpretive procedures are to be undertaken. The...
-
Larry Culp Redefines Success at GE When Larry Culp became the 12th chief executive officer of General Electric, he was the first CEO to be brought in from outside the company, and GE desperately...
-
Write a balanced equation for the ionization of nitric acid in water.
-
Predict the products of each precipitation reaction. Balance the equation, and then write the net ionic equation. (a) Pb(NO3)2(aq) (b) Ca(NO3)2(aq) (c) Ca(NO3)2(aq) + KBr(aq) + KF(aq) + NaCO4 (aq)
-
Is it possible to have proton pumping in the absence of electron transport?
-
Ford Motor Company (2023) reported that as of December 31, 2023, it had cash and cash equivalents of $24,862 million and a receivables account after allowances of $ 15,601 million. The receivables...
-
Business tax senario: XYZ corporation made a profit of $ 5 0 0 , 0 0 0 . However, after business expenses and deductions, the taxable income is $ 3 0 0 , 0 0 0 . Calculate the corporation's tax...
-
On August 1 , 2 0 2 2 , Colombo Company s treasurer signed a note promising to pay $ 1 2 2 , 1 0 0 on December 3 1 , 2 0 2 2 . The proceeds of the note were $ 1 1 5 , 8 0 0 . b . Calculate the...
-
For the experiment of rolling an ordinary pair of dice, find the probability that the sum will be less than 4 or greater than 6. The probability that the sum will be less than 4 or greater than 6 is...
-
What are at least 5 assets of the Air Force Security Forces organization that are at risk; that is, harm to one or more of the assets would impact the organization's ability to function normally. Be...
-
On January 1, 2011, Peres Company purchases 80% of the common stock of Soap Company for $308,000. Soap has common stock, other paid-in capital in excess of par, and retained earnings of $50,000,...
-
Flicker, Inc., a closely held corporation, acquired a passive activity this year. Gross income from operations of the activity was $160,000. Operating expenses, not including depreciation, were...
-
Propose a plausible mechanism for the following transformation: ['] ELOH
-
Devise an efficient synthesis for the following transformation (recall that aldehydes are more reactive than ketones): H.
-
Treatment of catechol with formaldehyde in the presence of an acid catalyst produces a compound with molecular formula C 7 H 6 O 2 . Draw the structure of this product. HO Catechol
-
Explain one way compensation professionals can strengthen the pay-for-performance link? What the strengths of this way? And what are the drawbacks?
-
Discuss anything you learned about personal financial management in Kindergarten through 12th grade. What do you remember? Has anyone in your life discussed financial management with you? If so, what...
-
Discuss how the business make decisions in the context of the behaviour of the various cost of production? Required ( have to be included) : Communicating of cost behaviour from accountant to...

Study smarter with the SolutionInn App


