The enthalpy change for the oxidation of styrene, C 8 H 8 , is measured by calorimetry.
Question:
The enthalpy change for the oxidation of styrene, C8H8, is measured by calorimetry.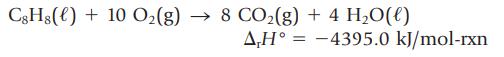
Use this value, along with the standard enthalpies of formation of CO2(g) and H2O(ℓ), to calculate the enthalpy of formation of styrene, in kJ/mol.
Transcribed Image Text:
C8Hg() + 10 O₂(g) → 8 CO₂(g) + 4H₂O(l) AH-4395.0 kJ/mol-rxn
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
To calculate the enthalpy of formation of styrene C8H8 i will use the enthalpy change for th...View the full answer

Answered By

Ashington Waweru
I am a lecturer, research writer and also a qualified financial analyst and accountant. I am qualified and articulate in many disciplines including English, Accounting, Finance, Quantitative spreadsheet analysis, Economics, and Statistics. I am an expert with sixteen years of experience in online industry-related work. I have a master's in business administration and a bachelor’s degree in education, accounting, and economics options.
I am a writer and proofreading expert with sixteen years of experience in online writing, proofreading, and text editing. I have vast knowledge and experience in writing techniques and styles such as APA, ASA, MLA, Chicago, Turabian, IEEE, and many others.
I am also an online blogger and research writer with sixteen years of writing and proofreading articles and reports. I have written many scripts and articles for blogs, and I also specialize in search engine
I have sixteen years of experience in Excel data entry, Excel data analysis, R-studio quantitative analysis, SPSS quantitative analysis, research writing, and proofreading articles and reports. I will deliver the highest quality online and offline Excel, R, SPSS, and other spreadsheet solutions within your operational deadlines. I have also compiled many original Excel quantitative and text spreadsheets which solve client’s problems in my research writing career.
I have extensive enterprise resource planning accounting, financial modeling, financial reporting, and company analysis: customer relationship management, enterprise resource planning, financial accounting projects, and corporate finance.
I am articulate in psychology, engineering, nursing, counseling, project management, accounting, finance, quantitative spreadsheet analysis, statistical and economic analysis, among many other industry fields and academic disciplines. I work to solve problems and provide accurate and credible solutions and research reports in all industries in the global economy.
I have taught and conducted masters and Ph.D. thesis research for specialists in Quantitative finance, Financial Accounting, Actuarial science, Macroeconomics, Microeconomics, Risk Management, Managerial Economics, Engineering Economics, Financial economics, Taxation and many other disciplines including water engineering, psychology, e-commerce, mechanical engineering, leadership and many others.
I have developed many courses on online websites like Teachable and Thinkific. I also developed an accounting reporting automation software project for Utafiti sacco located at ILRI Uthiru Kenya when I was working there in year 2001.
I am a mature, self-motivated worker who delivers high-quality, on-time reports which solve client’s problems accurately.
I have written many academic and professional industry research papers and tutored many clients from college to university undergraduate, master's and Ph.D. students, and corporate professionals. I anticipate your hiring me.
I know I will deliver the highest quality work you will find anywhere to award me your project work. Please note that I am looking for a long-term work relationship with you. I look forward to you delivering the best service to you.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
With reference to the "Chemistry Put to Work" box on explosives, (a) Use bond enthalpies to estimate the enthalpy change for the explosion of 1.00 g of nitroglycerin. (b) Write a balanced equation...
-
The standard enthalpies of formation of ClO and ClO2 are 101 and 102 kJ/mol, respectively. Using these data and the thermodynamic data in Appendix C, calculate the overall enthalpy change for each...
-
Use Table 8.4 to estimate the enthalpy change for each of the following reactions: a. H2C == O (g) + HCl (g) H3C - O - Cl (g) b. H2O2 (g) + 2CO (g) H2 (g) + CO2 (g) (c). 3H2C == CH2 (g) C6H12 (g)...
-
2. Using the data below, create the project schedule using normal times. Determine the order in which you would crash the project one day, two days, and so on until it is in an all-crash mode....
-
Describe the three types of technologies. Explain the strategic role of technology.
-
Suppose the following demand exists for iPhone apps: (a) At $9, what quantity is demanded? (b) If the price drops to $6, what quantity is demanded? (c) Is demand elastic or inelastic in that price...
-
If a valve in a pipe is suddenly closed, a large pressure surge may develop. For example, when the electrically operated shutoff valve in a dishwasher closes quickly, the pipes supplying the...
-
Lauren Tarson and Michele Progransky opened Top Drawer Optical seven years ago with the goal of producing fashionable and affordable eyewear. Tarson and Progransky have been very pleased with their...
-
An object moving along a horizontal track collides with and compresses a light spring (which obeys Hooke's Law) located at the end of the track. The spring constant is 40.7 N/m, the mass of the...
-
The following terms are used extensively in thermodynamics. Define each and give an example. (a) Exothermic and endothermic (b) System and surroundings (c) Specific heat capacity (d) State function...
-
The enthalpy change for the oxidation of naphthalene, C 10 H 8 , is measured by calorimetry. Use this value, along with the standard enthalpies of formation of CO 2 (g) and H 2 O(), to calculate the...
-
For 2022, assume that Wm Morrison Supermarkets plc (GBR) had pension expense of 61 million and contributed 52 million to the pension fund. Prepare Morrisons journal entry to record pension income and...
-
Distinguish between magical realism and surrealism. How do these genres differ in their approach to blending the fantastical with reality? Can you give examples of authors or works that epitomize...
-
How has the concept of the Bildungsroman evolved over time? Discuss contemporary variations of this genre and examine how they address themes of personal growth in different cultural or...
-
Describe how the arrangement of atoms allows for electrical current to move through metals? Explain.
-
what ways do metafictional techniques complicate the relationship between the author, reader, and narrative? provide examples from well-known metafictional works and analyze their effect on...
-
i. ii. iii. iv. Write a program/assembly code to evaluate the arithmetic statement: P = (Q * ((R+ (S* T)) - (U/V))) Using a general register computer with zero address instructions. Using a general...
-
What does the income statement report?
-
An item of depreciable machinery was acquired on 1 July 2009 for $120,000 by cash It is expected to have a useful life of 10 years and zero salvage value On 1 July 2012, it was decided to revalue the...
-
The vapor pressure of a liquid can be written in the empirical form known as the Antoine equation, where A(1), A(2), and A(3) are constants determined from measurements: Starting with this equation,...
-
Use the following vapor pressures of propane given here to calculate the enthalpy of vaporization using a graphical method or a least squares fitting routine. P (Torr) T (K) 0.01114 100. 120 2.317...
-
The vapor pressure of liquid benzene is 20,170 Pa at 298.15 K, and Î H vaporization =30.72 kJ mol -1 at 1 atm pressure. Calculate the normal and standard boiling points. Does your result for...
-
Razor USA produces a variety of electric scooters. Assume that Razor has just received an order from a customer (Pulse Cycles) for 750 Power Core scooters. The following price, based on cost plus a...
-
Your company has acquired Joggers PLC, a smaller company. The integration of the information systems can take up to 6 months, and until then Joggers PLC workers will continue following their...
-
The purpose of understanding social norms and conflict within teams is to help team members and team leaders understand how to optimize performance through awareness and intervention (when...

Study smarter with the SolutionInn App


