What mass of Na 2 CO 3 , in grams, is required for complete reaction with 50.0
Question:
What mass of Na2CO3, in grams, is required for complete reaction with 50.0 mL of 0.125 M HNO3?
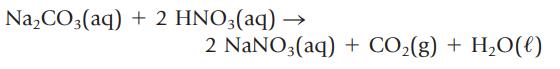
Transcribed Image Text:
Na₂CO3(aq) + 2 HNO3(aq) — 2 NaNO3(aq) + CO₂(g) + H₂O(l)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 40% (5 reviews)
To determine the mass of Na2CO3 required for complete reaction with 500 mL of 0125 M HNO3 ...View the full answer

Answered By

Navashree Ghosh
I believe in quality work and customer satisfaction. So, I can assure you that you will get quality work from me when you hire me. Let's work together and build a long-term association.
4.90+
82+ Reviews
116+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
1. What mass of H2 should be produced by the reaction of Al with 75.0 mL of 2.95M HCl? 2Al(s) + 6HCl(aq) 2AlCl3(aq) + 3H2 (g). ln the lab, 0.15g H2 was collected. What is the % yield of the...
-
Like many metals, aluminum reacts with a halogen (here the orange-brown liquid Br 2 ) to give a metal halide, aluminum bromide. (The white solid on the lip of the beaker at the end of the reaction is...
-
S sold to B a car for P1M. The condition for the sale is for B to execute a chattel mortgage on the thing sold. The balance of 800,000 is payable in twenty equal installments. B defaulted on his 2 or...
-
Assume that n is a positive integer. For each of the following algorithm segments, how many times will the innermost loop be iterated when the algorithm segment is implemented and run? 1) for k:=1 to...
-
Discuss the key differences between common and assignable causes of variation. Give examples.
-
How do the measured densities of a body compare at rest and in motion?
-
The stream function for an incompressible, two-dimensional flow field is \[ \psi=a y-b y^{3} \] where \(a\) and \(b\) are constants. Is this an irrotational flow? Explain.
-
Kayser Company owns 25% of Fort Company. For the current year Fort reports net income of $180,000 and declares and pays a $50,000 cash dividend. Record Kaysers equity in Forts net income and the...
-
Select a company you are familiar with and detail one short-term and one longer-term strategy, then discuss how the role of human resource development can provide valuable advice and counsel in the...
-
When an electric current is passed through an aqueous solution of NaCl, the valuable industrial chemicals H 2 (g), Cl 2 (g), and NaOH are produced. What mass of NaOH can be formed from 15.0 L of 0.35...
-
What volume of 0.109 M HNO 3 , in milliliters, is required to react completely with 2.50 g of Ba(OH) 2 ? 2 HNO3(aq) + Ba(OH)(s) - 2 HO(l) + Ba(NO3)2(aq)
-
Person A tosses a coin and then person B rolls a die. This is repeated independently until a head or one of the numbers 1, 2, 3,4 appears, at which time the game is stopped. Person A wins with the...
-
a. Equipment depreciation was $1,500. b. $700 of advertising expense was incurred but not paid. (Use Advertising Payable.) c. Office Supplies on hand at the end of the year totaled $250. The...
-
Is Google a monopoly? Why or why not? Research this issue.
-
If a comet nucleus partly vaporizes and loses material into the comet's head & tail each time its passes near the Sun, what eventually happens to the nucleus?
-
The velocity of sound is about 344 meters per second. What is the expected frequency of a recorder that is 44 cm long with all holes covered
-
Consider the rate of return of stocks ABC and XYZ . Year rABC rXYZ 1 2 3 % 3 8 % 2 8 1 2 3 1 7 1 8 4 5 1 5 3 1 3 _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ c ....
-
Davidson Hardware does not expect costs to change dramatically and wants to use a inventory costing method that averages cost changes. Requirements 1. Which inventory costing method would best meet...
-
a) Show that (a, b) := {{a}, {b}} does not satisfy the ordered pair axiom. b) Determine whether each of the following statements is true or false. (Give a reason in each case): (i) {a, b} C (a, b)....
-
Using toluene and acetylene as your only sources of carbon atoms, show how you would prepare the following compound.
-
Using the expression dS = C p /T dt VdP, calculate the decrease in temperature that occurs if 2.25 moles of water at 310. K and 1650. bar is brought to a final pressure of 1.30 bar in a reversible...
-
3.75 moles of an ideal gas with C V ,m = 3/2 R undergoes the transformations described in the following list from an initial state described by T = 298 K and P = 4.50 bar. Calculate q, w, U, H, and S...
-
1. Refer back to your StrengthsFinder results. How have you been able to use your Top 5 Strengths in a team environment (either in a class project or at work)? Please explain. 2. When you have worked...
-
Analysis of the Nature of change displayed by Nadler's (1995) model and insert where you believe Eagle Air is. Also include a discussion with regards to the differences between incremental and...
-
What is business strategy, corporate strategy, and corporate governance. 1) Explain why these strategies are important to us. 2) Reflect on the learning of the strategic management using your...

Study smarter with the SolutionInn App


