Consider a sample of an ideal gas with n and T held constant. Which of the graphs
Question:
Consider a sample of an ideal gas with n and T held constant. Which of the graphs below represents the proper relationship between P and V? How would the graph differ for a sample with a larger number of moles?
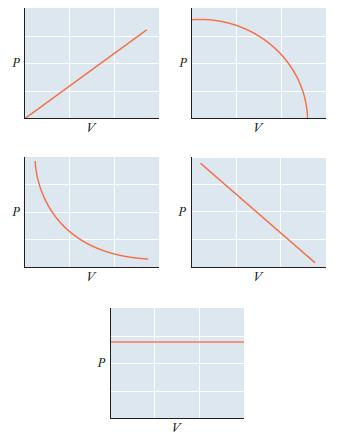
Transcribed Image Text:
P P V V P P P V V V
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
The graph on the left represents the proper relationship between P and V for a sample of an ideal ga...View the full answer

Answered By

Asim farooq
I have done MS finance and expertise in the field of Accounting, finance, cost accounting, security analysis and portfolio management and management, MS office is at my fingertips, I want my client to take advantage of my practical knowledge. I have been mentoring my client on a freelancer website from last two years, Currently I am working in Telecom company as a financial analyst and before that working as an accountant with Pepsi for one year. I also join a nonprofit organization as a finance assistant to my job duties are making payment to client after tax calculation, I have started my professional career from teaching I was teaching to a master's level student for two years in the evening.
My Expert Service
Financial accounting, Financial management, Cost accounting, Human resource management, Business communication and report writing. Financial accounting : • Journal entries • Financial statements including balance sheet, Profit & Loss account, Cash flow statement • Adjustment entries • Ratio analysis • Accounting concepts • Single entry accounting • Double entry accounting • Bills of exchange • Bank reconciliation statements Cost accounting : • Budgeting • Job order costing • Process costing • Cost of goods sold Financial management : • Capital budgeting • Net Present Value (NPV) • Internal Rate of Return (IRR) • Payback period • Discounted cash flows • Financial analysis • Capital assets pricing model • Simple interest, Compound interest & annuities
4.40+
65+ Reviews
86+ Question Solved
Related Book For 

Chemistry For Engineering Students
ISBN: 9780357026991
4th Edition
Authors: Lawrence S. Brown, Tom Holme
Question Posted:
Students also viewed these Sciences questions
-
(1 point) Write the system of equations as a matrix equation, that is, rewrite it in the form Input your answer below: a11 a12 a13 = a21 = a22= = a23 = a31 b3 = a32 = a33 = b = = b2 = || || - -2x +...
-
Consider 2.00 moles of an ideal gas that is taken from state A (PA = 2.00 atm, VA = 10.0 L) to state B (PB = 1.00 atm, VB = 30.0 L) by two different pathways. These pathways are summarized in the...
-
In 1858, J. Waterston found a clever way to estimate molecular sizes from macro- scopic properties of a liquid, by comparing its surface tension and heat of vaporiza- tion. The surface tension of...
-
A firm has total debt of $6,000,000 and stockholder's equity is $4,000,000. The firm wants to calculate equity-to- total asset ratio in order to make decision about further raise of capital. What is...
-
Palmer Corporation operates on a calendar-year basis. It begins the annual budgeting process in late August when the president establishes targets for the total dollar sales and net income before...
-
The following refers to units processed in Sunflower Printings binding department in March. Compute the total equivalent units of production with respect to labor for March using the weighted-average...
-
Mary Kay Morrow began working for Hallmark in 1982. At the beginning of 2002, Hallmark adopted the Hallmark Dispute Resolution Program, which required, among other things, that claims against the...
-
At December 31, 2012, Rivera Corporation reported the following plant assets During 2013, the following selected cash transactions occurred.Apr. 1 Purchased land for $2,200,000.May 1 Sold equipment...
-
3. Design NAND-NAND implementation for following functions: a) F1(x, y, z) = (x + y + z)(x' + y' + z') b) F2(x, y) = xy + x'y
-
Consider a sample of N 2 gas under conditions in which it obeys the ideal gas law exactly. Which of these statements is true? (a) A sample of Ne(g) under the same conditions must obey the ideal gas...
-
Why do heavier gases move more slowly than light gases at the same temperature?
-
How does reading comprehension on small, mobile devices compare with comprehension on full-size screens?
-
What were the underlying reasons for the dissatisfaction of American colonists with the system of mercantilism imposed by the British crown?
-
Licorne Inc. is considering a project which would involve the purchase of new manufacturing equipment at a cost of $8,657. The equipment would be depreciated on a straight-line basis to a book value...
-
Provide the full citations for the two most important Commonwealth Acts dealing with income tax. Provide the full citation for the Act and the section of that Act that grants power to the...
-
What's the link between federalism and individual liberty? How federalism conceives individual liberty?
-
Bruno carries on a cement business as a sole proprietor. He estimates the business will generate income of $100,000 during the taxation year ending December 31, 2022. If he incorporates the business,...
-
Compare and contrast at least three work related characteristics of Hispanic and Anglo-American cultures.
-
A handrail, which weighs 120 N and is 1.8 m long. was mounted to a wall adjacent to a small set of steps (Figure P4.26). The support at A has broken, and the rail has fallen about the loose bolt at 8...
-
An ideal gas sample containing 1.75 moles for which C V ,m = 5/2R undergoes the following reversible cyclical process from an initial state characterized by T = 275 K and P = 1.00 bar: a. It is...
-
For protein denaturation, the excess entropy of denaturation is defined as is the transition excess heat capacity. The way in which δC trs P can be extracted from differential scanning...
-
The standard entropy of Pb(s) at 298.15 K is 64.80 J K -1 mol - 1 . Assume that the heat capacity of Pb(s) is given by The melting point is 327.4C and the heat of fusion under these conditions is...
-
A 15 lb baby sits on the floor. Draw a free body diagram and label all of the acting forces. What is the normal force?
-
A joint taxpayer files their return with three (3) children. Both of the taxpayers have ITINs, but all of the children have SSNs. The return is prepared with a refund including Child Tax Credit,...
-
A particular type of UV radiation has a wavelength of 189 nm. Calculate the frequency of this radiation in s-1. Enter your answer with no units.

Study smarter with the SolutionInn App


