A 25.0-mL sample of 1.44 M NH 3 is titrated with 1.50 M HCl. Calculate the pH
Question:
A 25.0-mL sample of 1.44 M NH3 is titrated with 1.50 M HCl. Calculate the pH at the equivalence point.
Choose an indicator from Table 16.4, and justify your choice.
Table 16.4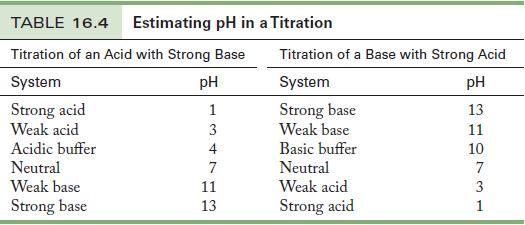
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
pH at the e...View the full answer

Answered By

Arun kumar
made more than four thousand assignments
5.00+
3+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
A 10.0-mL solution of 0.300 M NH3 is titrated with a 0.100 M HCl solution. Calculate the pH after the following additions of the HCl solution: (a) 0.0 mL, (b) 10.0 mL, (c) 20.0 mL, (d) 30.0 mL, (e)...
-
Calculate the pH at the halfway point and at the equiva-lence point for each of the following titrations. a. 100.0 mL of 0.10 M HC7H5O2 (Ka = 6.4 105) titrated with 0.10 M NaOH b. 100.0 mL of 0.10 M...
-
Please write detailed roadmap/solution for all questions below. 1) An output of nmap search is shown below, a) Type the required terminal command and required parameters to obtain the shown output....
-
How do recruitment and selection practices contribute to high performance in an organization?
-
Predict the products of the following nucleophilic acyl substitutionreactions: (b) (a) NaOH NH3 H20 H (d) (c) Na* "OCH3 C CH3NH2 SCH3 "CH
-
A 2-kW baseboard electric resistance heater in a vacant room is turned on and kept on for 15 min. The mass of the air in the room is 75 kg, and the room is tightly sealed so that no air can leak in...
-
The optimum speeds (in kilometers per hour) for 30 hatchbacks Approximate the mean of the frequency distribution. Car Speeds (in kilometers per hour) 20-24 Frequency 8 25-29 16 30-34 5 35-39 1
-
Noble Corporation has outstanding $400,000 of 8 percent bonds callable at 104. On December 1, immediately after the payment of the semiannual interest and the amortization of the bond discount were...
-
An ASTM A572 Grade 50 steel bar is attached to a rigid wall at one end and a spring at the other end, with the other end of the spring being attached to a rigid wall, as shown below. The bar is 20 cm...
-
Exactly 50 mL of a 0.0500 M solution of ethylamine, a base with K b = 1.1 10 -6 , is titrated with 0.100 M HNO 3 . What is the pH at the equivalence point? Suggest a good indicator from Table 16.4...
-
Chloropropionic acid, ClCH 2 CH 2 COOH, is a weak monoprotic acid with Ka = 7.94 10 -5 . Calculate the pH at the equivalence point in a titration of 10.00 mL of 0.100 M chloropropionic acid with...
-
In Exercises 4554, rationalize the denominator. 5 V3 - 1
-
Outline the importance of each of the following in an internal control system for cash: a. That all cash should be banked daily, intact b. The bank reconciliation statement c. The segregation of...
-
The bookkeeper at Covington Ltd undertakes a bank reconciliation at the end of every month. On 31 August, the bank reconciliation showed a deposit in transit of $650 and two outstanding cheques (no....
-
AAA Limited received its bank statement for the month ending 30 June, and reconciled the statement balance to the 30 June balance in the Cash account. The reconciled balance was determined to be...
-
List five recommendations for communicating successfully in a diverse and inclusive workplace. Be prepared to discuss each.
-
Peakhurst Limited had the following trial balance at 1 January 2016: You are given the following additional information for the year ended 31 December 2016: a. Bad debts of $8000 were written off. b....
-
What are the objectives of measures used to evaluate the financial performance of investment centers?
-
A copper rod of length L =18.0 in is to be twisted by torques T (see figure) until the angle of rotation between the ends of the rod is 3.08. (a) If the allowable shear strain in the copper is 0.0006...
-
What size of standard hydraulic copper tube from Appendix G.2 is required to transfer 0.06 m 3 /s of water at 80C from a heater where the pressure is 150 kPa to an open tank? The water flows from the...
-
Determine the required size of new Schedule 80 steel pipe to carry water at 160F with a maximum pressure drop of 10 psi per 1000 ft when the flow rate is 0.5 ft 3 /s.
-
A device designed to allow cleaning of walls and windows on the second floor of homes is similar to the system shown in Fig. 11.20. Determine the velocity of flow from the nozzle if the pressure at...
-
Have you found mnemonic devices or creative ways of remembering other mathematical or statistical concepts
-
Who is the functional sponsor for SPOT-ES and should be contacted if you have any questions regarding DoD OCS policies?
-
Describe the four stages in a digital forensic investigation. How does the Fourth Amendment and Fifth Amendment apply to digital forensics investigations? Provide at least one example of how the...

Study smarter with the SolutionInn App


