Using Table 9.4, calculate the energy required to break all of the bonds in one mole of
Question:
Using Table 9.4, calculate the energy required to break all of the bonds in one mole of the following compounds.
(a) NH3
(b) CH3OH
Table 9.4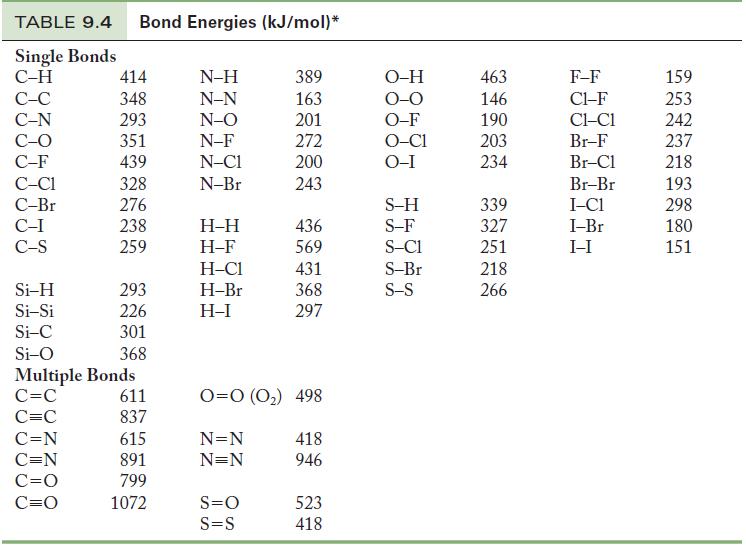
Transcribed Image Text:
TABLE 9.4 Bond Energies (kJ/mol)* Single Bonds C-H C-C C-N C-O C-F C-C1 C-Br C-I C-S Si-H Si-Si Si-C Si-O 414 348 293 351 439 328 276 238 259 C=N C=N C=O C=O 293 226 301 368 Multiple Bonds C=C C=C 611 837 615 891 799 1072 N-H N-N N-O N-F N-C1 N-Br H-H H-F H-C1 H-Br H-I 389 163 201 272 200 243 S=O S=S 436 569 431 368 297 0=0 (0₂) 498 N=N N=N 418 946 523 418 O-H O-O O-F O-C1 O-I S-H S-F S-C1 S-Br S-S 463 146 190 203 234 339 327 251 218 266 F-F C1-F CLC1 Br-F Br-Cl Br-Br I-C1 I-Br I-I 159 253 242 237 218 193 298 180 151
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
a 1...View the full answer

Answered By

Ajay Negi
Hi, I've completed my degree in engineering (Information Technology) from an NIT. Currently working as a software engineer. Wish to impart quality education to the future generation.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
Using Table 9.4, calculate the energy required to break all of the bonds in one mole of the following compounds. (a) CH 2 CF 2 (b) N 2 H 4 Table 9.4 TABLE 9.4 Bond Energies (kJ/mol)* Single Bonds...
-
The specific heat capacity of silver is 0.24 JoC-1g-1. a. Calculate the energy required to raise the temperature of 150.0 g Ag from 273 K to 298 K. b. Calculate the energy required to raise the...
-
Calculate the energy required to assemble the array of charges shown in Figure P25.34, where a = 0.200 m, b = 0.400 m, and q = 6.00 +C.
-
Why does the cost to load in an unconstrained system is lower compared to contrained with N - 1 Contingency in PLEXOS Modelling? What are the factors that affect the increase in cost to load during...
-
Below are listed the atomic weight, density, and atomic radius for three hypothetical alloys. For each determine whether its crystal structure is FCC, BCC, or simple cubic and then justify your...
-
Miller & Miller Company recorded sales, cost of goods sold, and ending inventory for the cur-rent year in the following amounts: $650,000, $500,000, and $128,000, respectively. Calculate the amount...
-
Ki Corp. is in liquidation under Chapter 7 of the Federal Bankruptcy Code. The bankruptcy trustee has established a new set of books for the bankruptcy estate. After assuming custody of the estate,...
-
Suppose Ramirez Ltd. Just issued a dividend of $1.89 per share on its common stock. The company paid dividends of $1.47, $1.62, $1.67, and $1.78 per share in the last four years. If the stock...
-
How do organizational change theories such as Lewin's Three-Stage Model and Kotter's Eight-Step Process adapt to accommodate the complexities of globalized, digitally-enabled business environments ?
-
Write the Lewis structures of HNNH and H 2 NNH 2 . Predict which molecule has the greater NN bond energy.
-
Which molecule has the most polar bond: N 2 , BrF, or ClF? Use an arrow to show the direction of polarity in each bond.
-
As the U.S. population ages, the number of people needing medical care increases. Unless a cure is found in the next decade, one of the most expensive diseases requiring such care is Alzheimers, a...
-
01.) Positive charges of +Q and negative charges of -Q are placed on the corners of a square as shown. What is the direction of the electric field E and the sign of the electric potential V at point...
-
9. 10. 11. A pith ball has a charge of -1.30 x 10-6 C. How many extra electrons are there? 8.13 x 102 e- A pith ball has a charge of 7.4 10-8 C. How many extra particles are there and what kind? 4.6...
-
08.) If the connecting wires and the battery in the circuit have negligible resistance, what is the current through the 6-ohm resistor? (A) 1/10 A (B) 1/3 A (C) 1/2 A (D) 1 A (E) 3/2 A Holo 6V 79...
-
Consider the figure with a symmetrical arrangement, in which qa = qb= +6.55 C, qc = 9d = -6.55 C, and q is positive. Using the symmetry of the arrangement, determine the direction of the force on...
-
5. In 2009, the world's fastest (and possibly coolest) man, Usain Bolt, set a world record for a 100 meter sprint with a time of 9.58 seconds. Round all answers to two decimal points in this problem....
-
Multiple Choice 1. An upward-sloping supply curve shows that a. Buyers are willing to pay more for particularly scarce products. b. Suppliers expand production as the product price falls. c....
-
Read the Forecasting Supply Chain Demand Starbucks Corporation case in your text Operations and Supply Chain Management on pages 484-485, then address the four questions associated with the...
-
The solid shaft is fixed to the support at C and subjected to the torsional loadings. Determine the shear stress at points A and B on the surface, and sketch the shear stress on volume elements...
-
The solid 30-mm-diameter shaft is used to transmit the torques applied to the gears. Determine the absolute maximum shear stress in the shaft. 300 N-m 500 N-m 200 N-m 400 N-m 300 mm B. 400 mm 500 mm
-
The solid aluminum shaft has a diameter of 50 mm. Determine the absolute maximum shear stress in the shaft and sketch the shear-stress distribution along a radial line of the shaft where the shear...
-
Routers can communicate with each other through dynamic or static routing. A dynamic routing protocol, such as protocol, such as (RIP, OSPF, BGP, and so on) learns about its directly connected routes...
-
Provide a simple C++ program that utilizes atomic operations. (The program should revolve around thread management) ?
-
In the Security section of Azure Services, which Azure Service can be used to protect Azure resources from denial of service threats?

Study smarter with the SolutionInn App


