Write the chemical equation and use the data in Tables 15.6 and 15.8 to calculate the base
Question:
Write the chemical equation and use the data in Tables 15.6 and 15.8 to calculate the base ionization constant for the following ions.
(a) Chlorite ion
(b) Fluoride ion
Table 15.6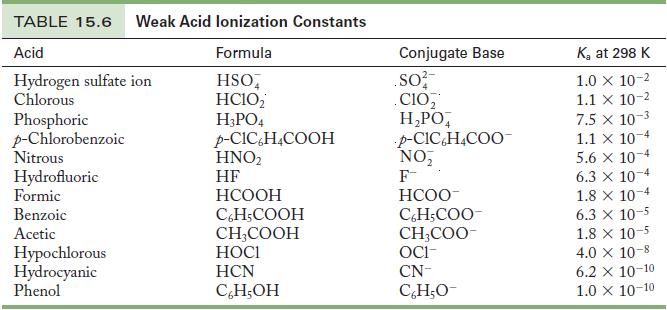
Table 15.8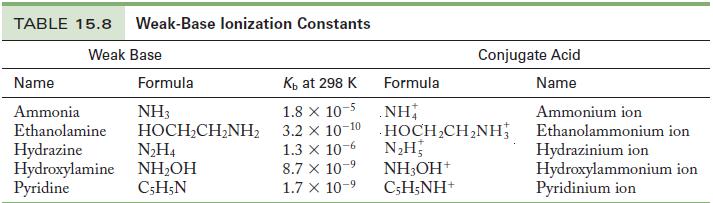
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
The base ionization constant K can be calculated using the formula Kb Kw Wh...View the full answer

Answered By

Joseph Mwaura
I have been teaching college students in various subjects for 9 years now. Besides, I have been tutoring online with several tutoring companies from 2010 to date. The 9 years of experience as a tutor has enabled me to develop multiple tutoring skills and see thousands of students excel in their education and in life after school which gives me much pleasure. I have assisted students in essay writing and in doing academic research and this has helped me be well versed with the various writing styles such as APA, MLA, Chicago/ Turabian, Harvard. I am always ready to handle work at any hour and in any way as students specify. In my tutoring journey, excellence has always been my guiding standard.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
Calcium acetate, Ca(CH 3 CO 2 ) 2 (aq), is used to treat patients with a kidney disease that results in high levels of phosphate ions in the blood. The calcium binds to the phosphates so that they...
-
Is the criterion 6 3CO 2 ) 2 (aq) is found to be 9.11. However, the contribution to the pH from the autoprotolysis of water was ignored. Repeat the calculation of the pH of this solution, taking into...
-
Although there are extensive tables available for the pK a of weak acids, you might be dealing with an unknown acid or a known acid at an unlisted temperature. You could then use a procedure like...
-
(10%) Problem 10: Suppose A = (-3.67 m)i + (4.57 m)j, B = (2.52 m)i + (-4.17 m)j + (2.39 m)k, and D= (-2.89 m)i + (-5.99 m)j. 50% Part (a) What is the angle, in degrees, between D and A? 0=11547...
-
The floor space of any facility that houses hell-and-tube heat exchangers must be sufficiently large so the tube bundle can be serviced easily. A rule of thumb is that the floor space must be at...
-
An air-standard Carnot cycle is executed in a closed system between the temperature limits of 350 and 1200 K. The pressures before and after the isothermal compression are 150 and 300 kPa,...
-
In a game of cards you are dealt a hand of six cards at random from a standard deck of 52 cards. You are declared winner if you are being dealt an ace, two kings and three queens. Find the...
-
The following cash flows were reported by Techno Inc. in 2009 and 2008. (a) Explain the difference between net income and cash flow from operating activities for Techno in 2009. (b) Analyze Techno...
-
On January 2, Year 1, a motorcycle dealer sells a motorcycle with an extended warranty for two years beyond the manufacturer's two-year warranty and an agreement to service the vehicle for the first...
-
Write the chemical equation and use the data in Tables 15.6 and 15.8 to calculate the acid ionization constant for the following ions. (a) Hydroxylammonium ion (b) Ammonium ion Table 15.6 Table 15.8
-
Write the chemical equation and use the data in Tables 15.6 and 15.8 to calculate the base ionization constant for the following ions. (a) Formate ion (b) Nitrite ion Tables 15.6
-
Which school of ethical thought is described in each of the following independent scenarios? Justify your choices. a. Julie and Bart were talking over their progress in the accounting program at Big...
-
Which of these is an important reason to review your writing and speaking choices carefully to make sure you are not using biased language? a. Biased language often reflects biased thinking, and it...
-
In terms of business etiquette, how can you reconcile the fact that mobile devices are now essential communication tools for most professionals with the fact that their use in meetings, over meals,...
-
How do your typeface selections help determine the personality of your documents and messages?
-
Revise the following sentences, using shorter, simpler words: a. Our superannuated content management system has proven to be ineffectual once again. b. It is imperative that the pay increments be...
-
Why are email, texting, and other forms of digital communication so prone to inadvertent etiquette breakdowns, in which even well-intentioned writers insult or confuse readers?
-
Describe goalpost conformance.
-
In the circuit shown in Figure 4, a battery supplies a constant voltage of 40 V, the inductance is 2 H, the resistance is 10, and l(0) = 0. (a) Find l(t). (b) Find the current after 0.1s.
-
Write a Verilog model for a 74HC192 synchronous 4-bit up/down counter. Ignore all timing data. Your code should contain a statement of the form always @(DOWN,UP, CLR, LOADB).
-
Consider the following 8-bit bidirectional synchronous shift register with parallel load capability. The notation used to represent the input/output pins is explained as follows: CLR Asynchronous...
-
Complete the following Verilog code to implement a counter that counts in the following sequence: Q = 1000, 0111, 0110, 0101, 0100, 0011, 1000, 0111, 0110, 0101, 0100, 0011, (repeats). The counter...
-
Treasury securities are now one of the best investments because of their high yields and default free. Currently, treasury bills (1, 3, 6, and 1 year) yield above 5% while 10 year notes and 30 year...
-
Due to the nature of their asset and liability contracts, depository institutions are the Financial Institutions most exposed to liquidity risk. What types of Financial Institutions have relatively...
-
Discussion Prompt: Profitability Ratios NIKE Return on Assets 8.4% Asset Turnover 1.0 % Gross Margin 43.8 Return on Common Shareholders' Equity 17.40% UNDER ARMOR Return on Assets (1.12) Asset...

Study smarter with the SolutionInn App


