Write the chemical equation for the reaction whose energy change is the standard enthalpy of formation of
Question:
Write the chemical equation for the reaction whose energy change is the standard enthalpy of formation of each of the following substances.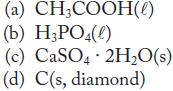

Transcribed Image Text:
(a) CH₂COOH(e) (b) H₂PO4(0) (c) CaSO4 2H₂O(s) (d) C(s, diamond)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
a CH3COOHl The standard enthalpy of formation of acetic acid ...View the full answer

Answered By

ANDREW KIPRUTO
Academic Writing Expert
I have over 7 years of research and application experience. I am trained and licensed to provide expertise in IT information, computer sciences related topics and other units like chemistry, Business, law, biology, biochemistry, and genetics. I'm a network and IT admin with +8 years of experience in all kind of environments.
I can help you in the following areas:
Networking
- Ethernet, Wireless Airmax and 802.11, fiber networks on GPON/GEPON and WDM
- Protocols and IP Services: VLANs, LACP, ACLs, VPNs, OSPF, BGP, RADIUS, PPPoE, DNS, Proxies, SNMP
- Vendors: MikroTik, Ubiquiti, Cisco, Juniper, HP, Dell, DrayTek, SMC, Zyxel, Furukawa Electric, and many more
- Monitoring Systems: PRTG, Zabbix, Whatsup Gold, TheDude, RRDtoo
Always available for new projects! Contact me for any inquiries
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
(a) Why are tables of standard enthalpies of formation so useful? (b) What is the value of the standard enthalpy of formation of an element in its most stable form? (c) Write the chemical equation...
-
Part 1 a. Ammonia, NH 3 , is a weak electrolyte. It forms ions in solution by reacting with water molecules to form the ammonium ion and hydroxide ion. Write the balanced chemical reaction for this...
-
Hydrogen iodide is a reactive compound that is sometimes used in the manufacture of methamphetamine. It can be produced by the direct reaction of the elements. Calculate the standard Gibbs free...
-
4. Apple Inc. is a US-based technology company that plans to invest in a new research and development center in Europe and aims to raise the required funds as follows: < < - - A bond issue of...
-
A precipitation process for producing perfect spheres of silica has been developed. The individual particles are so small that most cannot be discerned by the naked eye. Using optical microscopy, the...
-
Find the most general antiderivative or indefinite integral. You may need to try a solution and then adjust your guess. Check your answers by differentiation. fra. x (x + 1) dx
-
Mcfadzean and Honarzad run a farmers' market stall as a partnership. Honarzad withdrew \($26\) 300 cash on 25 October 2025. Mcfadzean withdrew \($17\) 500 on 28 November 2025. On 1 April 2026...
-
On July 1, 2016, Roland Company exchanged 18,000 of its $45 fair value ($1 par value) shares for all the outstanding shares of Downes Company. Roland paid acquisition costs of $40,000. The two...
-
4. The atomic numbers of elements, U, V, W and X are 13, 14, 16 and 17 respectively. Which one of the elements will react with oxygen to form an ionic compound? (Atomic number of oxygen is 8) X A. B....
-
Using a linear programming framework and the data from case Exhibit 1, formulate and solve the problem of finding the cost-minimizing set of bids that meet Mars' business requirements and determines...
-
Use standard enthalpies of formation to calculate the enthalpy change for each of the following reactions at 298.15 K and 1 atm. Label each as endothermic or exothermic. (a) The fermentation of...
-
Write the formation reaction for each of the following substances. (a) HBr(g) (b) HSO4(l) (c) 03(g) (d) NaHSO4(s)
-
Evaluate the definite integral. Use a graphing utility to verify your result. So x(x - 2) dx
-
Show that if the ratio of the specific heats is 1.4 , then \[\left(\frac{\partial p}{\partial T} ight)_{s}=\frac{7}{2}\left(\frac{\partial p}{\partial T} ight)_{v}\]
-
An air-standard Diesel cycle operates with a compression ratio, \(r=10: 1\). If the initial conditions at bdc are \(1 \mathrm{bar}\) and \(27^{\circ} \mathrm{C}\), and the energy addition is \(2000...
-
For a van der Waals gas that obeys the state equation \[p=\frac{R T}{v-\mathrm{b}}-\frac{\mathrm{a}}{v^{2}}\] shows that the coefficient of thermal expansion, \(\beta\), is given by \[\beta=\frac{R...
-
What has the development of the basic Rankine cycle carried out in Problems P3.9-P3.14 shown you about the effect of the salient parameters on the efficiency of the cycle? Evaluate the mean...
-
The operating processes of a spark-ignition engine can be represented by the Otto cycle, which is internally reversible and gives a thermal efficiency of \[\eta_{\text {Otto...
-
True or false? Explain. a. The value of a share equals the discounted stream of future earnings per share. b. The value of a share equals the PV of earnings per share assuming the firm does not grow,...
-
D Which of the following is considered part of the Controlling activity of managerial accounting? O Choosing to purchase raw materials from one supplier versus another O Choosing the allocation base...
-
Calculate H R and G R for the reaction AgNO 3 (aq) + KCl(aq) AgCl(s) + KNO 3 (aq).
-
Calculate the pH of a buffer solution that is 0.200 molal in CH 3 COOH and 0.15 molal in CH 3 COONa using the Davies equation to calculate . What pH value would you have calculated if you had...
-
Use the Davies equation to calculate for a 1.00 molar solution of KOH. Compare your answer with the values in Table 10.3. 0.2 m 0.7 m 0.4 m 0,8 m 0.9 m Substance 0.1 m 0.3 m 0.5 m 0.6 m 1.0 m 0.657...
-
You have a revolving credit account at an annual percentage rate of 18%. Use the average daily balance method to find the new balance given the following statement of account. June 1 Previous month's...
-
Suppose that call options on a stock with strike prices of $30 and $35 cost $6.9 and $4.1, respectively. Use these options to construct a bear spread ("\_). What is the profit of the strategy if the...
-
10-year US Treasury yields 2.60%. Benin sovereign credit spread is 2.50% above US Treasury. Benin quasi-sovereign credit spread is .50% above Benins sovereign spread. Benin BigBank, a private...

Study smarter with the SolutionInn App


