Which arrangement of cations (yellow) and anions (blue) in a lattice is the more stable? Explain your
Question:
Which arrangement of cations (yellow) and anions (blue) in a lattice is the more stable? Explain your reasoning.
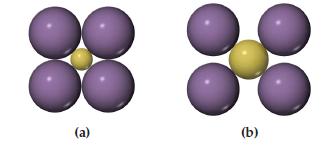
Transcribed Image Text:
(a) (b)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
Recall that similar charges have strong repulsive tendencies against ea...View the full answer

Answered By

Vijesh J
My passion to become a tutor is a lifetime milestone. Being a finance and marketing professional with hands-on experience in wealth management, portfolio management, team handling and actively contributing in promoting the company. Highly talented in managing and educating students in most attractive ways were students get involved. I will always give perfection to my works. Time is the most important for the works and I provide every answer on time without a delay. I will proofread each and every work and will deliver a with more perfection.
4.70+
5+ Reviews
15+ Question Solved
Related Book For 

Chemistry The Central Science
ISBN: 9780321910417
13th Edition
Authors: Theodore E. Brown, H. Eugene LeMay, Bruce E. Bursten, Catherine Murphy, Patrick Woodward, Matthew E. Stoltzfus
Question Posted:
Students also viewed these Sciences questions
-
Explain why anions are always larger than the atoms from which they are derived, whereas cations are always smaller than the atoms from which they are derived.
-
Consider the two stereo isomers of 3-isoproplcyclohexanol. (a) Which is the more stable conformation of each stereo isomer? (b) Which is the more stable stereo isomer by how much?
-
Name the following compound, identify each substituent as axial or equatorial, and tell whether the conformation shown is the more stable or less stable chair form (yellow-greenC1):
-
Accounts Receivable AED 5,000 Accounts Payable 15,000 Advertising Expense 2,000 16,500 Building Cash Common Stock 50,000 Dividends 1,200 Equipment 2,000 Land 70,000 Notes Payable 60,000 3,400 Office...
-
Oxygen at 100 kPa and 200oC is compressed to half its initial volume. Determine the final state in terms of pressure (p2) and temperature (T2) if the compression is carried out in an (a) Isobaric....
-
Try this in the bathtub or when youre washing dishes: Lower a drinking glass, mouth downward, over a small floating object such as a Ping-Pong ball. What do you observe? How deep must the glass be...
-
On a cool day in April a scantily clothed runner is known to lose heat at a rate of \(450 \mathrm{~W}\) when running on a level surface because of convection to the surrounding air at...
-
Bolling Electronics manufactures DVD players for commercial use. W. Blaker Boiling, president of Bolling Electronics, is contemplating producing DVD players for home use. The activities necessary to...
-
Calculate and report the classification accuracy for the given confusion matrix. 215 0 2 0 5 2 224 0 135 34 0 2 40 211 0 16 368 1 0 12 397 1 0 2 458 0 0 461 3 0 1 20 183 183 30 237 36 219 o 187 419...
-
1. Do you think that only certain individuals are attracted to these types of jobs, or is it the characteristics of the jobs themselves that are satisfying? 2. What characteristics of these jobs that...
-
The electronic structure of a doped semiconductor is shown here. (a) Which band, A or B, is the valence band? (b) Which band is the conduction band? (c) Which band consists of bonding molecular...
-
Two solids are shown below. One is a semiconductor and one is an insulator. Which one is which? Explain your reasoning.
-
In the middle of the night you are standing a horizontal distance of 14.0 m from the high fence that surrounds the estate of your rich uncle. The top of the fence is 5.00 m above the ground. You have...
-
A large population is in Hardy-Weinberg equilibrium at a locus A with two alleles, A and a. The frequency of the A allele is p. Assuming that the population mates at random with respect to the A...
-
What are the intricate mechanisms underlying the hypothalamic-pituitary-adrenal axis and its role in stress response regulation ?
-
Report Outline Scenario (SC) I want a full report on the below topic To write pages of a report based on the outline- Class Scenario University of Manitoba is dealing with complaints from students...
-
short paragraph explanation is enough. You want to start a kit-meal delivery business like Hello Fresh. Macro factors : Identify and explain how any external (macro) factors would impact your...
-
Read each of the situations presented below and then determine what barrier or barriers were present. For each situation, identify the barrier or barriers and then state what should the communicators...
-
Datacom Services Inc. reported the following statement of changes in shareholders' equity for the year ended October 31, 2017. Requirements Answer these questions about Datacom Services's...
-
A heat engine has a heat input of 3 Ã 104 Btu/h and a thermal efficiency of 40 percent. Calculate the power it will produce, in hp. Source 3 x 10 Btu/h 40% HE Sink
-
How does a trigonal pyramid differ from a tetrahedron so far as molecular geometry is concerned?
-
Describe the bond angles to be found in each of the following molecular structures: (a) Planar trigonal, (b) Tetrahedral, (c) Octahedral, (d) Linear.
-
(a) What is meant by the term electron domain? (b) Explain in what way electron domains behave like the balloons in Figure 9.5.Why do they do so? Two balloons linear orientation Three balloons...
-
Janice will need to pay $200 at the end of every month for the next 12 months, except for the payment of the 8th month. What is the present value, assuming a rate of 4%, compounded quarterly?
-
List and explain two management tools in the planning process and two measurable performance indicators. Explain in detail.
-
What does a high PE tell us about the value of the stock price (over or under valued)? What does a low PE tell us about the value of the stock price (over or under valued)? Be specific with your...

Study smarter with the SolutionInn App


