Use the bond energies in Table 17.1 and the accounting format shown in Section 17.5 to determine
Question:
Use the bond energies in Table 17.1 and the accounting format shown in Section 17.5 to determine whether these reactions are exothermic or endothermic:
H2 + Cl2 ¡ 2 HCl
2 HC‚CH + 5 O2 → 4 CO2 + 2 H2O
Table 17.1
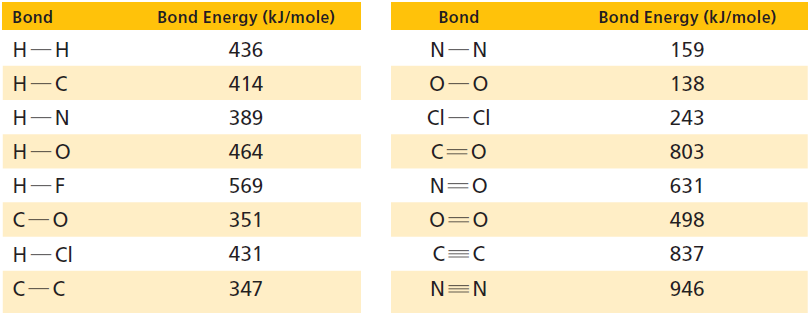
Transcribed Image Text:
Bond Energy (kJ/mole) Bond Energy (kJ/mole) Bond Bond Н-Н 436 159 Н-С 414 138 Cl-CI 389 Н—N 243 803 Н—О 464 C=0 Н-—F 569 N=0 631 351 498 Н— СІ 431 837 N=N 347 946
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (9 reviews)
a Energy to break bonds Energy released from bond formation H x H 43...View the full answer

Answered By

Amos Kiprotich
I am a wild researcher and I guarantee you a well written paper that is plagiarism free. I am a good time manager and hence you are assured that your paper will always be delivered a head of time. My services are cheap and the prices include a series of revisions, free referencing and formatting.
4.90+
15+ Reviews
21+ Question Solved
Related Book For 

Conceptual Physical Science
ISBN: 978-0134060491
6th edition
Authors: Paul G. Hewitt, John A. Suchocki, Leslie A. Hewitt
Question Posted:
Students also viewed these Physics questions
-
Use the average bond energies in Table 4.3 to estimate ÎU for the reaction C 2 H 4 (g) + H 2 (g) C 2 H 6 (g). Also calculate ÎU o R from the tabulated values of ÎH o f for reactant...
-
The following table summarizes 204 endothermic reactions involving sodium bicarbonate. Let A denote the event that a reactions final temperature is 271 K or less. Let B denote the event that the heat...
-
2 Section 3 Date 4567 8 9 10 11 12 13 14 15 16 STRANK 21 22 23 Pharoah Inc. sells storage buildings of various sizes to homeowners and businesses. Respond to the requirements related to the following...
-
apple company: 1.what are key characteristics of the industry? 2. where is the company in its life cycle?
-
Arthur Wesson, an unmarried individual who is age 68, reports 2014 taxable income of $160,000. He records AMT positive adjustments of $40,000 and tax preferences of $35,000. a. What is Arthur's AMT?...
-
The following is the statement of financial position of WW Associates as at 31 December 2008: During 2009, the following transactions took place: 1 The owners withdrew equity in the form of cash of...
-
For each of the following questions appearing on an internal accounting control questionnaire, assume that a "No" answer is given: 1. Is there separation of duties between the journalizing of...
-
Presented below are the trial balance and the other information related to Yorkis Perez, a consulting engineer. 1. Fees received in advance from clients $6,000.2. Services performed for clients that...
-
On January 1, Boston Company completed the following transactions (use a 7% annual interest rate for all transactions): ( FV of $1, PV of $1, FVA of $1, and PVA of $1) (Use the appropriate factor(s)...
-
CHEMISTRY SINGLE REPLACEMENT REACTION WORKSHEET 1. Use the Activity Series Table to determine if a single replacement reaction occurs. if it does, move to step 2. If it does not occur write NR (no...
-
Rank the following in order of increasing number of atoms: (a) 52 g of vanadium, V; (b) 52 g of chromium, Cr; (c) 52 g of manganese, Mn.
-
Bakers yeast contains a biological catalyst known as catalase, which catalyzes the transformation of hydrogen peroxide, H 2 O 2 , into oxygen, O 2 , and water, H 2 O. Write a balanced equation for...
-
Kenisha Johnson, CEO of Eastern Electronics Inc., is looking at the possibility of marketing a new product line. Kenisha will be approaching a risk capital investor, Manon Miller, asking for $500,000...
-
Youre the director of corporate communications and the employee newsletter is produced by your office. Today you receive this email message from Tonya Freira: Write a response to Tonya Freira. Also,...
-
You work for a local fitness center called Super Fit. The owners of Super Fit would like to expand their business and add a running track and a large pool. They have drafted the following letter to...
-
Evaluate the following subject lines. Is one subject line in each group clearly best? Or does the best line depend on company culture, whether the message is a paper memo or an email message, or on...
-
Rewrite the following negative message so it follows the guidelines for negative messages: Dear Valued Employee: I'm afraid the company will not be able to grant you your requested vacation time at...
-
Three days ago, one of your subordinates forwarded to everyone in the office a bit of email humor hed received from a friend. Titled You know youre Southern when . . . , the message poked fun at...
-
What has helped drive the increasing awareness of the importance of business ethics and social responsibility?
-
Velshi Printers has contracts to complete weekly supplements required by fortysix customers. For the year 2018, manufacturing overhead cost estimates total $600,000 for an annual production capacity...
-
Liquid nitrogen at 77 K is stored in a cylindrical container having an inside diameter of 25 cm. The cylinder is made of stainless steel and has a wall thickness of 1.2 cm. Insulation is to be added...
-
The Fourier field equation in cylindrical coordinates is a. What form does this equation reduce to for the case of steady-state, radial heat transfer? b. Given the boundary conditions T = T i at r =...
-
Perform the same operations as in parts (a), (b), and (c) of Problem 16.1 with respect to a spherical system. Data From Problem 16.1 The Fourier field equation in cylindrical coordinates is a. What...
-
What is the balance in Work in Process Inventory at the end of each month? Work in Process Inventory April 30 $ May 31 A 10,140 15,950 June 30 $ 10,450
-
Sako Company s Audio Division produces a speaker used by manufacturers of various audio products. Sales and cost data on the speaker follow: Selling price per unit on the intermediate market $ 6 0...
-
Vulcan Company's contribution format income statement for June is as follows: Vulcan Company Income Statement For the Month Ended June 30 Sales Variable expenses Contribution margin Fixed expenses...

Study smarter with the SolutionInn App


