A pure fluid is described by the Antoine equation With P in bar and T in kelvin.
Question:
A pure fluid is described by the Antoine equation
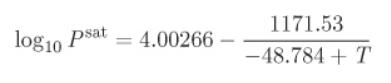
With P in bar and T in kelvin. The density of the liquid at room temperature is 0.656 g/cm3. How much heat is required to evaporate 1 kg of that liquid at 25 °C?
Transcribed Image Text:
log 10 psat 4.00266- = 1171.53 -48.784 + T
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
To calculate the heat required to evaporate 1 kg of the liquid at 25 C you can use the heat of vaporization equation Q m H Where Q Heat required in Jo...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Fundamentals Of Chemical Engineering Thermodynamics
ISBN: 9780132693066
1st Edition
Authors: Themis Matsoukas
Question Posted:
Students also viewed these Engineering questions
-
In this problem, we want to create a spreadsheet to help someone estimate their income in retirement. To do this, we will have them enter several numbers: the amount of money currently in savings,...
-
A fluid formed by pentane and hexane obeys Raoults law quite well. Such a mixture can be used for example as the working fluid of an organic Rankine cycle power plant. The vapor pressure of pure...
-
A pure fluid is described by the canonical equation of state: G = ( T ) + RT ln P, where (T ) is a substance-specific function of temperature. Determine for such a fluid expressions for V, S, H, U,...
-
Which of the following do ergonomics most directly address? Physical wellbeing Mental wellbeing Emotional wellbeing Social wellbeing
-
Consider a market in which Bert from Problem 4 is the buyer and Ernie from Problem 5 is the seller. a. Use Ernies supply schedule and Berts demand schedule to find the quantity supplied and quantity...
-
What factors might an individual investor take into account in determining his or her investment policy?
-
Which of the three components of Figure 1.2 might be a source of concern regarding "ethics"? Products Management Decisions Information Customer Transactions Data Analyze Measure, the + Categorize...
-
Peerless Windows is a major manufacturer and installer of windows into new homes. Currently, management has found that it has experienced a large number (about 15% of all orders placed) of customer...
-
1. What are some specific factors that make countries unattractive to firms looking to do business there? 2. What are some of the factors that firms must consider before deciding to begin...
-
A 3 m 3 insulated tank contains steam at 1 bar, 150 C. The tank is connected to a steam line that is maintained at 10 bar, 300 C until the pressure in the tank is 5 bar. How much steam (kg) was...
-
Oxygen is compressed from 1 bar, 25 C, to 150 bar. To avoid overheating, compression is done in stages followed by intercooling with water at 30 C so that the temperature exiting each stage of the...
-
Our definition of a group is somewhat broad. Would you classify each of the following collections of people as a group? Explain why or why not. a. 70,000 people at a football game. b. Students taking...
-
It is May 5, 2014. The quoted price of a government bond with a \(12 \%\) coupon that matures on July 27, 2024, is \(110-17\). What is the cash price?
-
You are the maker of a note in which you undertake to pay $500 to a moneylender on demand. Subsequently you pay an installment of $200 to the moneylender on this liability. What steps should you take...
-
Wilma purchased a used minivan, including a three-month warranty on parts and labour, from Xenon Used Cars Inc. for $10 000. She paid $1500 down and signed a promissory note for $8500, payable in...
-
It is January 9, 2015. The price of a Treasury bond with a \(12 \%\) coupon that matures on October 12, 2030, is quoted as 102-07. What is the cash price?
-
For decades, each winter Timson has openly entered the fields of an uninhabited farm adjoining his home and cross-country skied several times a week. He knows the Abel family that used to farm there,...
-
Redo the cash budget in Exhibit 24.1. a. Assume that the desired minimum level of cash is $4,000 instead of $10,000. What is the change in the firms need for funds? b. Assume that 60 percent of sales...
-
7. Baladna wants to analyze process that includes delivery by suppliers, production inside the company, transportation to to its customers and information systems. Then it also wants to find out...
-
A single vaporliquid equilibrium point for the water (1) + ethanol (2) system is experimentally measured at 30C. The experiment provides the following information: x 1 = 0.30, y 1 = 0.23, and P =...
-
You are part of a team evaluating the separation of a mixture of ethanol (1) + butyl methyl ether (2). This was a project your company explored a few years earlier. In that project, team members...
-
For an equimolar mixture of n-hexane (1) + benzene (2) at 150C and 5 bar, estimate the molar volume of the mixture three ways: A. Ideal gas law B. van der Waals equation of state C. Peng-Robinson...
-
Assume you notice the following information. Assume you spend $1 million USD do an arbitrage trading strategy. What is your profit is USD . Remember to consider the profit after you pay back your...
-
A 20-year bond with a coupon rate of 9%, payable semiannually, has an early redemption option after 5 years with a price of $1,050. The bond is sold at a yield to maturity rate of 8%. What is the...
-
MV Corporation has debt with market value of $95 million, common equity with a book value of $95 million, and preferred stock worth $20 million outstanding. Its common equity trades at $49 per share,...

Study smarter with the SolutionInn App


