Estimate the standard enthalpies of formation at 25 C and 1 bar of (a) OH(g); (b) N
Question:
Estimate the standard enthalpies of formation at 25 °C and 1 bar of
(a) OH(g);
(b) N2H4(g).
Write Lewis structures and use data from Table 10.3, as necessary.
Table 10.3
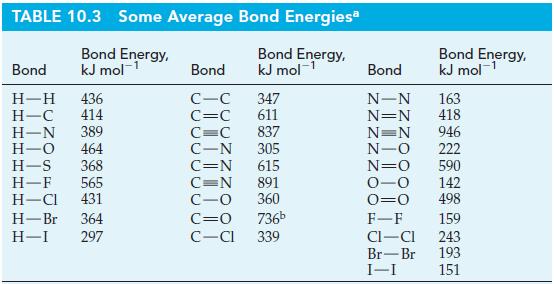
Transcribed Image Text:
TABLE 10.3 Some Average Bond Energiesa Bond Energy, kJ mol-¹ Bond Energy, kJ mol-¹ Bond H-H 436 H-C 414 H-N 389 H-O 464 H-S 368 H-F 565 H-Cl 431 H-Br 364 H-I 297 Bond C-C C=C C=C 837 C-N 305 C=N 615 347 611 C=N C-O C=O 736b C-Cl 339 891 360 Bond Energy, kJ mol-¹ 1 Bond N-N 163 N=N 418 N=N 946 -0 222 N=O 590 -O 142 0=0 498 F-F CI-CI Br-Br I-I 159 243 193 151
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
12H2g 12028 AH AH AH rxn OHs AH bonds brokenAH b...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
The standard enthalpies of formation of S(g), F(g), SF4(g), and SF6(g) are 1278.8 kJ/ mol, 179.0 kJ/ mol, 775 kJ/ mol, and 1209 kJ/ mol, respectively. a. Use these data to estimate the energy of an...
-
The standard free energies of formation and the standard enthalpies of formation at 298 K for difluoroacetylene (C2F2) and hexafluorobenzene (C6F6) are For the following reaction: C6F6(g) 3C2F2(g) a....
-
The standard enthalpies of formation of ClO and ClO2 are 101 and 102 kJ/mol, respectively. Using these data and the thermodynamic data in Appendix C, calculate the overall enthalpy change for each...
-
In each of the homeowners forms, OA) the property coverage is the same OB) the liability coverage varies OC) both the property and liability coverage are the same OD) the property coverage varies
-
Discuss and explain whether there is valid consideration for each of the following promises: (a) A and B entered into a contract for the purchase and sale of goods. A subsequently promised to pay a...
-
What products would you expect from Hofmann elimination of the following amines? If more than one product is formed, indicate which ismajor. NH2 (b) NH2 (a) CH3CH2CH2CHCH2CH2CH2CH3 NHCH2CH3 (d) NH2...
-
In the months leading up to the 2016 election, Christopher Steele, a former British intelligence agent, was hired by a Washington, D.C., research firm to investigate whether then-candidate Donald...
-
Recording Bad Debts Sandel Company reports the following financial information before adjustments. Prepare the journal entry to record bad debt expense assuming Sandel Company estimates bad debts at...
-
Create a relational model for the following ERD. Please do not develop a graphical version of the relational databases. Please just list the tables, attributes, primary keys and foreign keys. Dept...
-
Two of the following have the same shape. Which two, and what is their shape? What are the shapes of the other two? NI 3 , HCN, SO 3 2- , NO 3 - .
-
One of the chemical reactions that occurs in the formation of photochemical smog is O 3 + NO NO 2 + O 2 . Estimate r H for this reaction by using appropriate Lewis structures and data from Table...
-
Consider the following mechanism for the hydrolysis of ethyl acetate. Write a complete balanced equation for the overall reaction, list any intermediates, and identify the catalyst in this reaction....
-
Explain the nature of the field of clinical psychology Provide a definition of clinical psychology, supported by course material. What is the scientific focus of the field and why that is important...
-
Read the articles, examine the advertisements, and peruse the pictures. Describe the audience that this magazine is targeting. Tell me as much about this audience as you can (age, sex, occupation,...
-
Select a company for which you believe its social capital both within the company and among its suppliers and customers is vital to its competitive advantage. Explain why you believe its social...
-
Match the term with its definition: A. Negative relevance B. "Plus" +s C. Credibility D. Documentation E. Facts 1. Statement that counts against another statement 2. Is used if two or more premises...
-
The managers at Point Products are uncertain what to do with these costs estimation results. Specifically, they are uncertain which, if either, method (account analysis or regression analysis) they...
-
On July 15, 2013, the Nixon Car Company purchased 1,000 tires from the Harwell Company for $50 each. The terms of the sale were 2/10, n/30. Nixon uses a periodic inventory system and the gross method...
-
Reichenbach Co., organized in 2018, has set up a single account for all intangible assets. The following summary discloses the debit entries that have been recorded during 2018 and 2019. Instructions...
-
Write down the formulas for an investments NPV and rate of return. Prove that NPV is positive only if the rate of return exceeds the opportunity cost of capital.
-
What is the net present value of a firms investment in a U.S. Treasury security yielding 5 percent and maturing in one year?
-
A parcel of land costs $500,000. For an additional $800,000 you can build a motel on the property. The land and motel should be worth $1,500,000 next year. Suppose that common stocks with the same...
-
Increasing cost of cat food are potential WHAT when reviewing a SWOT analysis for a nonprofit cat rescue, external threat, internal threat, external weakness, or internal weakness?
-
Yuri needs to fill an open departmental position. In what situation should Yuri use the nine-box grid?
-
What are some disadvantages to using debt to fund your assets?

Study smarter with the SolutionInn App


