Starting with 0.3500 mol CO(g) and 0.05500 mol COCl 2 (g) in a 3.050 L flask at
Question:
Starting with 0.3500 mol CO(g) and 0.05500 mol COCl2(g) in a 3.050 L flask at 668 K, how many moles of Cl2(g) will be present at equilibrium?
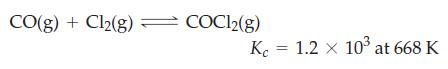
Transcribed Image Text:
CO(g) + Cl2(g) = COC1₂(g) Kc = 1.2 x 10³ at 668 K
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
1 Set up the ICE table Species Initial mol Change mol Equilibrium ...View the full answer

Answered By

Kalyan M. Ranwa
I have more than seven years of teaching experience in physics and mechanical engineering.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
(A) If 0.150 mol H 2 (g) and 0.200 mol I 2 (g) are introduced into a 15.0 L flask at 445 C and allowed to come to equilibrium, how many moles of HI(g) will be present? (B) Suppose the equilibrium...
-
In the contact process, sulfuric acid is manufactured by first oxidizing SO2 to SO3, which is then reacted with water. The reaction of SO2 with O2 is A 2.000-L flask was filled with 0.0400 mol SO2...
-
A balloon with a volume of 1.50 L is at a pressure of 760 torr and a temperature of 30C. If the pressure is increased to 2300 torr and the temperature is raised to 72C, what is the new volume of the...
-
1.What is the difference between Router and firewall 2.What is Packet filtering router 3.What is Stateful inspection firewall 4.What is Circuit level firewall 5.What is Application level firewall...
-
JWG Company publishes Creative Crosswords. Last year the book of puzzles sold for $10 with variable operating cost per book of $8 and fixed operating costs of $40,000. How many books must JWG sell...
-
Patrick Kellogg has prepared the following list of statements about the accounting cycle. 1. "Journalize the transactions" is the first step in the accounting cycle. 2. Reversing entries are a...
-
Assume that Fossil sold watches to a department store on account for \($43,000\). How would this transaction affect Fossils accounting equation? a. Increase both liabilities and stockholders equity...
-
Palmer Company uses an activity-based costing system. It has the following manufacturing activity areas; related drivers used as allocation bases, and cost allocation rates: During the month, 100...
-
Janessa prefers high-end cosmetics, but she also likes a good deal. She only purchases her make-up at department stores when they have a gift with purchase. Today she saw at Macy's that if she buys...
-
Equilibrium is established in a 2.50 L flask at 250 C for the reaction How many moles of PCl 5 , PCl 3 , and Cl 2 are present at equilibrium, if (a) 0.550 mol each of PCl 5 and PCl 3 are initially...
-
Starting with 0.280 mol SbCl 3 and 0.160 mol Cl 2 , how many moles of SbCl 5 , SbCl 3 , and Cl 2 are present when equilibrium is established at 248 C in a 2.50 L flask? SbC15(g) = SbCl3(g) + Cl(g) Kc...
-
The bookkeeper for Geronimo Company has prepared the following balance sheet as of July 31, 2008. The following additional information is provided. 1. Cash includes $1,200 in a petty cash fund and...
-
The Story of Apple: Apple Inc. is an American multinational technology company headquartered in Cupertino, California, that designs, develops, and sells consumer electronics, computer software, and...
-
Given: pH = -log [H+] If the pH is 6.5 what is [H+]? Give 3 significant figures
-
John, a Canadian citizen, and Maria, a citizen of Mexico, connected through an online international pen-pal program a year and a half ago. They began exchanging messages and developed a deep...
-
Offender A is a 20-year-old white male who is a senior business management major at Clayton State University. He is currently completing an internship with a prominent financial investment firm and...
-
What principles from this week's study can you apply in your life and your social work career?
-
A value chain for an airline such as Delta or American would typically include marketing and promotion, ticketing and reservations, airport operations, and aircraft operations. Required Outline the...
-
From 1970 to 1990, Sri Lanka's population grew by approximately 2.2 million persons every five years. The population in 1970 was 12.2 million people.What is the best formula for P, Sri Lanka's...
-
Colliers Company has determined that the variable overhead rate is $2.90 per direct labor hour in the Fabrication Department. The normal production capacity for the Fabrication Department is 14,000...
-
The following data relate to factory overhead cost for the production of 5,000 computers: If productive capacity of 100% was 8,000 hours and the factory overhead cost budgeted at the level of 5,000...
-
Perma Weave Textiles Corporation began January with a budget for 30,000 hours of production in the Weaving Department. The department has a full capacity of 40,000 hours under normal business...
-
During Heaton Company's first two years of operations, it reported absorption costing net operating income as follows: Year 1 Year 2 Sales (@ $62 per unit) $ 992,000 $ 1,612,000 Cost of goods sold (@...
-
Cranbrook Used Books experienced the following events during the current year: a. Incurred $12,000 in selling costs. b. Took an inventory at year-end and learned that goods costing $100,000 were on...
-
The following events occurred at Moore's Hobbies and Crafts store during the most recent fiscal year: a. Purchased merchandise costing $129,000. b. Had sales revenue for the year totaling $175,000;...

Study smarter with the SolutionInn App


