Comment on the modes of bonding of the ligands in the Mn(II) complexes listed at the end
Question:
Comment on the modes of bonding of the ligands in the Mn(II) complexes listed at the end of Section 21.8, drawing attention to any conformational restrictions.
Data from Section 21.8
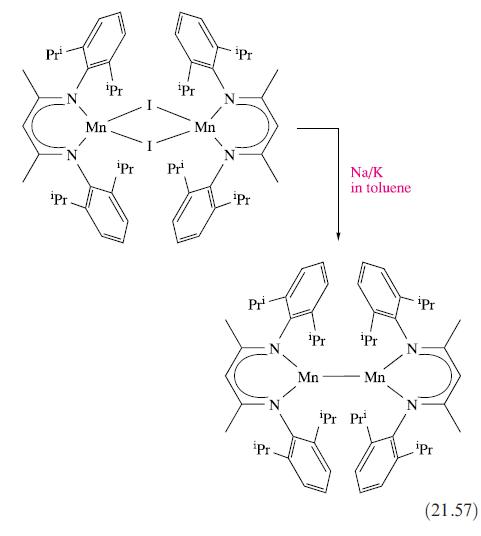
Transcribed Image Text:
Pr Pr iPr Mn iPr iPr Pr¹ Mn Pr ¹Pr Pr N iPr ¹Pr Mn Na/K in toluene Pr Mn iPr Pr Pr ipr (21.57)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 87% (8 reviews)
In order to comment on the modes of bonding of the ligands in the MnII complexes listed in equation 2157 we first need to understand the abbreviations ...View the full answer

Answered By

User l_1006857
I am a computer science professional with expertise in databases, AI programming, data structures and algorithms, and mathematics. With a strong background in these areas, I possess the knowledge and skills necessary to design and optimize database systems, develop intelligent algorithms and models, and solve complex computational problems. My proficiency in SQL, NoSQL, machine learning techniques, and mathematical concepts equips me to contribute to innovative projects and drive technological advancements.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
(a) Confirm that H 2 Os 3 (CO) 11 has sufficient valence electrons to adopt a triangular metal framework. Do the modes of bonding of the CO and H ligands affect the total valence electron count?...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-6. On December 12, Irene purchased the building where her store is located. She paid...
-
List and describe the payment options (terms of sale) that can be applied to domestic U.S. shipments.
-
The following represents a project that should be scheduled using CPM: a. Draw the network. b. What is the critical path? c. What is the expected project completion time? d. What is the probability...
-
A synchronous 4-bit UP/DOWN binary counter has a synchronous clear signal CLR and a synchronous load signal LD. CLR has higher priority than LD. Both CLR and LD are active high. D is a 4-bit input to...
-
Tennessee law imposes durational-residency requirements on persons and companies wishing to operate retail liquor stores, requiring applicants for an initial license to have resided in the state for...
-
(a) On January 1, 2012, Fishbone Corporation sold a building that cost $250,000 and that had accumulated depreciation of $100,000 on the date of sale. Fishbone received as consideration a $240,000...
-
A company is planning to purchase and store two items, gadgets and widgets. Each gadget costs $2.00 and occupies 2 square meters of floor space; each widget costs $3.00 and occu pies 1 square meter...
-
(a) Which of the following complexes would you expect to suffer from a JahnTeller distortion: [CrI 6 ] 4 , [Cr(CN) 6 ] 4 , [CoF 6 ] 3 and [Mn(ox) 3 ] 3 ? Give reasons for your answers. (b) [Et 4 N] 2...
-
Values of oct for [Ni(OH 2 ) 6 ] 2+ and high-spin [Mn(OH 2 ) 6 ] 3+ have been evaluated spectroscopically as 8500 and 21000 cm 1 respectively. Assuming that these values also hold for the...
-
Obtain a Thvenin equivalent for the circuit of Fig. 4-26 to the left of terminals ab. 10 2 10 2 h 10 2 Is.c. 5 b
-
What is one of your personal or academic goals? Using the ABCS approach, define how you will reach this goal. What stood out the most in your CSFI assessment? use beginning wording.
-
A permitting system for PRCS's involves: Preparation and Issuance of permits Permit use Cancellation and Safe Termination of the Entry and Permit Review of the Entry for continual improvement...
-
1. If you eat 750 calories of food and then run up several steps of stairs to work off the energy you taken in. How high do you climb if your mass is about 55kg? Assume 80% efficiency in the...
-
Using the following Two-Way Contingency Table, Find P(C) (Express as a decimal, round to 2 places if necessary) A B C X 0.09 0.35 0.13 Y 0.12 0.21 0.10
-
Solve 6. secx Stan 4 1 - tan x dx
-
Michael has always been overweight, and now he has decided to do something about it. He recently read in a news story that the IRS allows a medical expense deduction for the cost of certain weight...
-
The Adjusted Trial Balance columns of a 10-column work sheet for Webber Co. follow. Complete the work sheet by extending the account balances into the appropriate financial statement columns and by...
-
Identify the structure of the starting alkene in each of the following cases: a. b. c. 1) O3 C3H14 2) DMS C10H16 1) 03 2) DMS
-
When 2-methylpropane is treated with bromine in the presence of UV light, one product predominates. (a) Identify the structure of the product. (b) Draw the structure of the expected minor product....
-
Identify the most electronegative element in each of the following compounds: a) CH 3 OCH 2 CH 2 NH 2 b) CH 2 ClCH 2 F c) CH 3 Li
-
1 How many boards 6 in. wide will it take to cover a floor 259 in. wide? 6 1 boards 6 in. wide will cover a floor 259 in. wide. 6
-
Vision Inc. is considering leasing an equipment. The equipment costs $3,000,000 and it would be depreciated straight-line to zero over 4 years. It will be worthless in 4 years. Lease payment is...
-
1. What is a SQL Server? 2. Why is a SQL Server helpful? 3. Characteristics of a SQL Server? 4. Examples of how to use a SQL Server?

Study smarter with the SolutionInn App


