Determine G o (298 K) for the reaction: given the following data: What does the value of
Question:
Determine ΔGo(298 K) for the reaction:
![]()
given the following data:
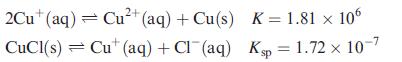
What does the value of ΔGo tell you about the tendency of precipitated CuCl to disproportionate?
Transcribed Image Text:
2+ 2CuCl(s) Cu²+ (aq) + 2Cl(aq) + Cu(s)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (4 reviews)
To determine Go standard Gibbs free energy change for the given reaction we can use the relationship ...View the full answer

Answered By

Surendar Kumaradevan
I have worked with both teachers and students to offer specialized help with everything from grammar and vocabulary to challenging problem-solving in a range of academic disciplines. For each student's specific needs, I can offer explanations, examples, and practice tasks that will help them better understand complex ideas and develop their skills.
I employ a range of techniques and resources in my engaged, interesting tutoring sessions to keep students motivated and on task. I have the tools necessary to offer students the support and direction they require in order to achieve, whether they need assistance with their homework, test preparation, or simply want to hone their skills in a particular subject area.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
What does the example of WorldCom tell you about the value of financial securities from different perspectives (Profit, Cash Flow, and Strategic Perspectives) as it relates to managerial decision...
-
Johnson Filtration, Inc., provides maintenance service for water filtration systems throughout southern Florida. Customers contact Johnson with requests for maintenance service on their water...
-
What does Descartes' rule of signs tell you about the number of positive real zeros and number of negative real zeros of the following function? g(x) = - x8 + 2x6 - 4x3 - 1
-
A restaurant records the following data over a month for its food: Opening inventory: $31,000 Purchases: $88,000 Closing Inventory: $28,000 Transfers in: $800 Transfers out: $200 Employee meals:...
-
How does a large-scale entry differ from a small-scale entry?
-
1. Jamie Lee is considering a used vehicle, but cannot decide where to begin her search. Using Your Personal Financial Plan Sheets #19, name the sources available to Jamie Lee for a used car...
-
On February 16, 2005, Joseph and Margaret Buset borrowed $192,000 from Fremont Investment & Loan (the Originator). The loan was secured by a mortgage on a residential condominium. The mortgage named...
-
Lazy River Resort opened for business on June 1 with eight air-conditioned units. Its trial balance before adjustment on August 31 is as follows. In addition to those accounts listed on the trial...
-
Problem 4. Consider the following imperfect information game. Notice that both play- ers have two information sets. 5,2 1 a b 2 t 1 d d 4,3 6.0 4,6 0,4 6,0 (a) Find all subgame perfect Nash...
-
Give the oxidation state of each element in the following compounds and ions; Pauling electronegativity values in Appendix 7 may be useful: (a) CaO; (b) H 2 O; (c) HF; (d) FeCl 2 ; (e) XeF 6 ; (f)...
-
Using data from Table 8.1 and from Section 8.3, explain why H 2 is evolved when powdered Ag is heated with a concentrated solution of HI. Data from Table 8.1 Table 8.1 Selected standard reduction...
-
Iman has the task of reviewing the evidence from substantive analytical procedures conducted by the audit associates on the audit of Smalley Services Inc. The audit associates have reported the...
-
List some of the reports you can generate with Project 2013 to assist with project time management.
-
Ace publishing company needs new equipment for a new branch in the region. According to prior estimates, the following positive cash flow will be generated during 5 years: Estimated pay-back period...
-
List the tools and techniques for performing risk control.
-
Why is quality assurance becoming more important? What does it mean to use lean in quality assurance?
-
What are the three main categories of outputs for quality control?
-
According to its 2013 annual report, RONA Inc. is the largest Canadian distributor and retailer of hardware, home renovation, and gardening products. RONAs 2013 financial statements contained the...
-
Interest Compounded Annually. When P dollars is invested at interest rate i, compounded annually, for t years, the investment grows to A dollars, where A = P(1 + i) t . Trevor's parents deposit $7800...
-
From a knowledge of their chemical properties, speculate on why cerium and europium were the easiest lanthanoids to isolate before the development of ion-exchange chromatography.
-
Neither lanthanoid nor actinoid organometallic compounds obey the 18-electron rule. Discuss the reasons, using the structures of the tris(Cp) and tris(Cp*) Ln and An complexes as examples.
-
Explain the variation in ionic radii between La 3+ and Lu 3+ .
-
Choose a company from the New York Stock Exchange that seems like it might be a good investment. Research the company, and also use Google Finance or the website of the New York Stock Exchange to...
-
Consider the following functions: f(x)=4-x g(x) = x+10x+16 1 h(x) = x+9 A) What are the domains of each function? Answer in interval notation. B) What is the domain of C) What is the domain of D(x) =...
-
Solve for x. 17x+7=15 -4x Round your answer to the nearest thousandth. Do not round any intermediate computations.

Study smarter with the SolutionInn App


