Rationalize the bond dissociation energy (D) and bond length data of the gaseous diatomic species given in
Question:
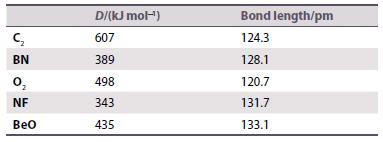
Rationalize the bond dissociation energy (D) and bond length data of the gaseous diatomic species given in the following table and highlight the atoms that obey the octet rule.
Transcribed Image Text:
C₂ BN NF BeO D/(kJ mol-¹) 607 389 498 343 435 Bond length/pm 124.3 128.1 120.7 131.7 133.1
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 25% (4 reviews)
To rationalize the bond dissociation energy D and bond length data for the gaseous diatomic species given in the table we first need to understand the ...View the full answer

Answered By

Deepankur Keserwani
0.00
0 Reviews
10+ Question Solved
Related Book For 

Inorganic Chemistry
ISBN: 9780198768128
7th Edition
Authors: Mark Weller, Tina Overton, Jonathan Rourke
Question Posted:
Students also viewed these Sciences questions
-
3/233 At the point A of closest approach to the sun, a comet has a velocity vA = 188,500 ft/sec. Deter- mine the radial and transverse components of its velocity vg at point B, where the radial...
-
Write Lewis structures that obey the octet rule for each of the following, and assign oxidation numbers and formal charges to each atom: (a) OCS, (b) SOCl2 (S is the central atom), (c) BrO3-, (d)...
-
The potential energy of two atoms in a diatomic molecule is approximated by V(r) = a/r12 - b/r2, where r is the spacing between atoms and a and b are positive constants. (a) Find the force F( r) on...
-
1. Consider the following problem: Suppose that the interest rate in this year is 5% and all investors expect that interest rates for the next 3 years will be as follows: Year 1 2 3 Forward Interest...
-
Consider the prices in the following three Treasury issues as of May 15, 2011: The bond in the middle is callable in February 2012. What is the implied value of the call feature? 6.500 May 17n 8.250...
-
We always see the same face of the Moon because the rotation of the Moon on its axis matches the rate at which it revolves around Earth. Does it follow that an observer on the Moon always sees the...
-
SWIGART v. BRUNO CALIFORNIA COURT OF APPEALS 13 CAL. APP. 5TH 529 2017 According to the American Endurance Ride Conference, endurance riding is a highly competitive and demanding sport. It is...
-
One subunit of Boxing Sports Company had the following financial results last month: Requirements 1. Complete the performance evaluation report for this subunit (round to two decimal places). 2....
-
4. Ethan bought new car worth $60 000. After 5 years, the car was worth $35 429.40. Calculate the depreciation rate of the Ethan's car. [T4] [C2]
-
In which of the species lCl 6 and SF 4 is the bond angle closest to that predicted by the VSEPR model?
-
Use the Ketelaar triangle in Fig. 2.28 and the electronegativity values in Table 1.7 to predict what type of bonding is likely to dominate in (a) BCl 3 , (b) KCl, (c) BeO. Figure 2.28. Table 1.7. 3 2...
-
In view of the uncertainty principle, why does a baseball seem to have a well-defined position and speed whereas an electron does not?
-
How do strategic leaders effectively synchronize organizational objectives with dynamic market forces to optimize long-term viability?
-
A square - section test piece is being used in a tensile test. The width and breadth of the test piece are each 15 mm. Calculate the stress in the test piece when a load of 3150 N is applied.
-
Assume the following economy produces only skateboards and the year 1981 is the base year. . Year Quantity Produced Price 1981 75 $30 1982 100 $33 1983 125 $38 1984 150 $42 What is the nominal GDP...
-
A force of 1 Newton will cause a mass of 1 kg to have an acceleration of 1 m/s 2 . Thus it follows that a force of 5 Newtons applied to a mass of 5 kg will cause it to acquire an acceleration of?...
-
What is the impact on the accounting if the company is not a going concern?
-
A telemarketer at a credit card company is instructed to ask the next 18 customers that call into the 800 number whether they are aware of the new Platinum card that the company is offering. Of the...
-
Nike manufactures shoes and sportswear. How has the Internet changed the way this company communicates with its suppliers and retail customers?
-
The diagrams at the right-hand side of Fig. 5.19 show three of the MOs in NH 3 . Sketch representations of the other four MOs. Figure 5.19. Energy 2p(a) 2p, 2p, (e) 2s (a) N e ta ## # NH C3v LGO(2)...
-
Titanium dissolves nitrogen to give a solid solution of composition TiN 0.2 ; the metal lattice defines an hcp arrangement. Explain what is meant by this statement, and suggest whether, on the basis...
-
Refer to Fig. 5.17 and the accompanying discussion. (a) Why does the B 2p z atomic orbital become a non-bonding MO in BH 3 ? (b) Draw schematic representations of each bonding and antibonding MO in...
-
A Nice Manager The management promotion process at Chisum Industries was a benchmark for providing lateral moves, as well as promotion to the next level within the company. With offices, plants, and...
-
Determine the values of the stress in portions AC and CB of the steel bar shown Fig. when the temperature of the bar is -45C knowing that a close fit exists at both of the rigid supports when the...
-
4.27 Determine the axial force in each bar of the plane truss loaded as shown in Fig. G. (Ans. S -S4+2 P; S2=-S6=-2.236 P S3=+P; Ss=0) 4.28 Determine the axial force in each bar of the plane truss...

Study smarter with the SolutionInn App


