Refer to Fig. 5.17 and the accompanying discussion. (a) Why does the B 2p z atomic orbital
Question:
Refer to Fig. 5.17 and the accompanying discussion.
(a) Why does the B 2pz atomic orbital become a non-bonding MO in BH3?
(b) Draw schematic representations of each bonding and antibonding MO in BH3.
Figure 5.17.
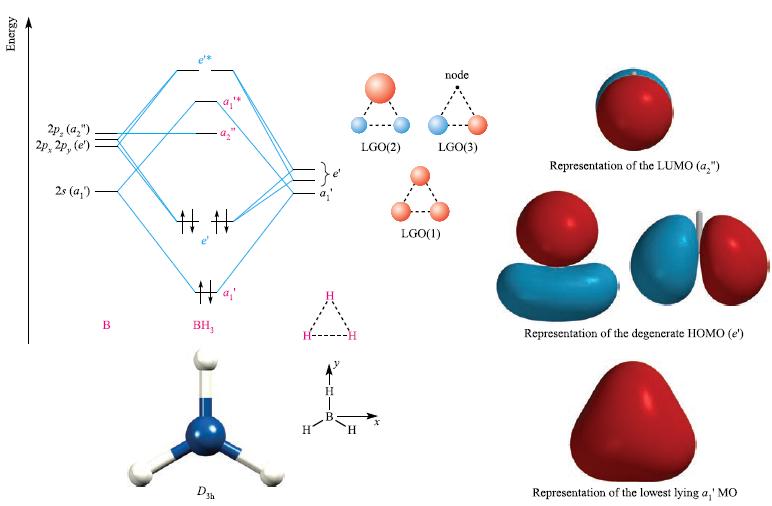
Transcribed Image Text:
Energy 2p, (a₂") 2p, 2p, (e) 2s (a₂) B # # Hai BH, LGO(2) node. LGO(3) LGO(1) Representation of the LUMO (a₂") Representation of the degenerate HOMO (e') Representation of the lowest lying a,' MO
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (6 reviews)
a In BH the B 2p and 2p atomic orbitals interact with the hydrogen 1s orbitals to form bonding and antibonding molecular orbitals because they are pro...View the full answer

Answered By

Fahmin Arakkal
Tutoring and Contributing expert question and answers to teachers and students.
Primarily oversees the Heat and Mass Transfer contents presented on websites and blogs.
Responsible for Creating, Editing, Updating all contents related Chemical Engineering in
latex language
4.40+
8+ Reviews
22+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Use a ligand group orbital approach to describe the bonding in [NH 4 ] + . Draw schematic representations of each of the bonding MOs.
-
Refer to Exhibit 14.8 and the accompanying discussion in the text. EXHIBIT 14.8 A Detailed Comparison of Actual Variable Costs and Flexible-Budget Variable Costs Demonstrate that the flexible-budget...
-
Refer to Exhibit 14.8 and the accompanying discussion in the text. EXHIBIT 14.8 A Detailed Comparison of Actual Variable Costs and Flexible-Budget Variable Costs Demonstrate that the materials usage...
-
State SQL query to satisfy the requirements below: Include the following columns: First Name Last Name Return all rows from the people table where the person's last name is between B and H. Use the...
-
After watching the video on snack foods in China, discuss the following: 1. What strategy are American companies employing in China? 2. How does Frito Lays approach impact their business? 3. Based...
-
What criteria are used by regulators to examine a thrift institution?
-
In the past five years, there have been significant innovations in technology such as smartphones and tablets. Technology companies rely on intellectual property (IP) rights, such as patents,...
-
Bumblebee Company estimates that 300,000 direct labor hours will be worked during the coming year, 2017, in the Packaging Department. On this basis, the following budgeted manufacturing overhead cost...
-
A jewelry shop specializes in creating gold and silver charms. The shop has five metalsmiths who work on the charms. One week, the shop's production possibilities curve shifts inward. What could...
-
Titanium dissolves nitrogen to give a solid solution of composition TiN 0.2 ; the metal lattice defines an hcp arrangement. Explain what is meant by this statement, and suggest whether, on the basis...
-
Table 5.6 gives the results of a self-consistent field (SCF) quantum chemical calculation for H 2 O using an orbital basis set of the atomic orbitals of O and the LGOs of an H---H fragment. The axis...
-
The parties, by and large, do not dispute the material facts. The Defendant, Phuong Le, is a 49-year-old who lived in Vietnam until 1991 . . . Le and his family, including his first wife, moved to...
-
CoffeeCarts has a cost of equity of 15.3%, has an effective cost of debt of 3.6%, and is financed 70% with equity and 30% with debt. What is this firms WACC?
-
Do callable bonds have a higher or lower yield than otherwise identical bonds without a call feature? Why?
-
When will the yield to maturity be higher than the yield to call for a callable bond?
-
ECB Co. has 1.45 million shares outstanding selling at $25 per share. It plans to repurchase 102,000 shares at the market price. What will its market capitalization be after the repurchase? What will...
-
Describe just-in-time inventory management.
-
Explain the basic differences between periodic and perpetual inventory systems.
-
Suppose you won a financial literacy competition and are given FJS10000 to invest, with the condition that investment can be done either in, i) Invest in Unit trust of Fiji or Invest in Fijian...
-
The effectiveness of platinum in catalysing the reaction 2H + (aq) + 2 e H 2 (g) is greatly decreased in the presence of CO. Suggest an explanation.
-
Tetramethyllead can be synthesized in the laboratory from lead(II) chloride and a Grignard reagent according to the following reaction: Assuming that the synthesis was attempted with 4.0 g of PbCl 2...
-
Alkanes are observed to exchange hydrogen atoms with deuterium gas over some platinum metal catalysts. When 3,3-dimethylpentane in the presence of D 2 is exposed to a platinum catalyst and the gases...
-
What investment decision rule should Canadian Tire Corporation use to analyze the firm's projects and make capital budgeting decisions for an Initial investment of $ 25,000,000.00?
-
what is the current yield on this, based on the closing price? bond is hwl 8 1/2 28 vol is 125,400 close is 67.75 net change is +7
-
Copperton Company issued bonds with a coupon rate of 8% (with half of the annual amount of interest paid every six months) and a face amount of $5,000. The bonds mature in 15 years. The market...

Study smarter with the SolutionInn App


