Use the following data to calculate average values of B(SeF) in SeF 4 and SeF 6 .
Question:
Use the following data to calculate average values of B(Se–F) in SeF4 and SeF6. Comment on your answers in view of the corresponding values for
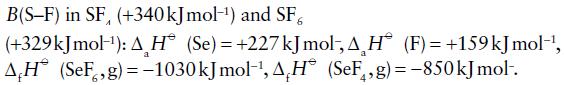
Transcribed Image Text:
B(S-F) in SF, (+340 kJ mol-¹) and SF a (+329kJmol-¹): AH (Se) = +227 kJ mol, A H (F)= +159 kJmol-¹, AH (SeF,g)=-1030 kJ mol-¹, AH (SeF,g)=-850 kJ mol-.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (7 reviews)
To calculate the average values of BSeF in SeF4 and SeF6 we can use the following thermodynamic data ...View the full answer

Answered By

User l_917591
As a Business Management graduate from Moi University, I had the opportunity to work as a tutor for undergraduate students in the same field. This experience allowed me to apply the theoretical knowledge I had gained in a practical setting, while also honing my teaching and communication skills.
As a tutor, I was responsible for conducting tutorial sessions, grading assignments and exams, and providing feedback and support to my students. I also assisted with the preparation of course materials and collaborated with other tutors and professors to ensure consistency in teaching and assessment.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Inorganic Chemistry
ISBN: 9780198768128
7th Edition
Authors: Mark Weller, Tina Overton, Jonathan Rourke
Question Posted:
Students also viewed these Sciences questions
-
Use the following data to calculate economic profit in year1. ear Sales Expenses other than interest Depreciation Earnings before interest and taxes (EBIT) Taxes on EBIT @40% Eamings before interest...
-
Use the following data to calculate Chiara's 2012 AMT base. Chiara files as a single taxpayer. Taxable income....................................$148,000 Positive AMT...
-
Use the following data to calculate Chiara's 2016 AMT base. Chiara files as a single taxpayer and itemizes her deductions. Taxable income .........................................$148,000 Positive...
-
A Styrofoam slab has thickness h and density ps. When a swimmer of mass m is resting on it, the slab floats in fresh water with its top at the same level as the water surface. Find the area of the...
-
Data for L. Takemoto, interior decorating, are presented in E2-2. Instructions Journalize the transactions using journal page J1.
-
Find a parametric representation for the surface. The part of the ellipsoid x 2 + 2y 2 + 3z 2 = 1 that lies to the left of the xz-plane
-
Mind & Motion entered into a real estate purchase contract (REPC) with Celtic Bank to buy a large piece of the banks foreclosed property. Although the county had approved plans to construct...
-
Consecutive five-year balance sheets and income statements of Donna Szabo Corporation are shown below and on the following page. Required a. Compute or determine the following for the years 20052009:...
-
Suppose that T: R R is a linear map, and . . . 2 2 3 T2 3 T1 = 2 and T 0 2 5 0 Then 2 T4 7
-
Use the bond enthalpy data in Table 2.7 to calculate the enthalpy of formation for NF 3 and NCl 3 . Explain why NF 3 is thermodynamically stable whereas NCl 3 is unstable and reactive. Table 2.7....
-
The elements are all metals. The most stable oxidation state at the top of the group is +3, the most stable at the bottom is +6. Identify the group of elements.
-
Using the letters E , I , J , L , and S to abbreviate the simple statements, Egypts food shortage worsens, Iran raises the price of oil, Jordan requests more U.S. aid, Libya raises the price of oil,...
-
If an interviewer asks you about a weakness, what is the best way to answer the question? If an interviewer asks for one or more letters of recommendation from your references, which of the following...
-
Deer Lodge Regional Hospital plans to use activity-based costing to assign hospital indirect costs to the care of patients. The hospital has identified the following activities and activity rates for...
-
For this Case Study Strategy and Design Assignment, you will propose a case study that could be used as the basis for a research project you might like to conduct at some point in time. Choose a...
-
In a short reflection paper, identify and analyze a past conflict that you have experienced and look at it from every angle. Include the following about your conflict: - How the conflict arose - How...
-
Hello! Assignemnt Leadership. Please answer with references! Question: The future of work is becoming more flexible. What are some of the challenges leaders may face with this new way of working? ...
-
Which of the following sets of quantum numbers are not allowed in the hydrogen atom? For the sets of quantum numbers that are incorrect, state what is wrong in each set. a. n = 3, = 2, m = 2 b. n =...
-
Willingness to pay as a measure of a person's value for a particular good measures the maximum a person would be willing to pay requires that payment actually be made depends on the satisfaction that...
-
The [Se 4 ] 2+ ion has D 4h symmetry and the SeSe bond lengths are equal (228 pm). (a) Is the ring in [Se 4 ] 2+ planar or puckered? (b) Look up a value of r cov for Se. What can you deduce about the...
-
Briefly discuss the trends in boiling points and values of vap H listed in Table 17.2 for the hydrogen halides. Table 17.2. Property Physical appearance at 298 K Melting point /K Boiling point/K...
-
For a given atom Y, the YF bond is usually stronger than the corresponding YCl bond. An exception is when Y is oxygen (Table 16.2). Suggest a reason for this observation. Table 16.2. 0-0 146 S-S 266...
-
https://act.pearsoncmg.com/activity/1/4/2 Beginning of Year: End of Year: Assets Liabilities $27,000 $18,000 $64,000 $26,000 1) What is the equity at the beginning of the year? Beginning Equity 9000...
-
Daniels Company made the following selected transactions during May: 1. Received cash from sale of stock, $55,000. 2. Paid creditors on account, $7,000. 3. Billed customers for services on account,...
-
MBA:8180 Managerial Finance Risk, Diversification, and Required Returns for Business Divisions T.A. Rietz, 2022 You just took an internship at Tooth-n-Hair, a private company that makes toothbrushes...

Study smarter with the SolutionInn App


