Using data in Appendix 11, determine which of the following species is thermodynamically unstable with respect to
Question:
Using data in Appendix 11, determine which of the following species is thermodynamically unstable with respect to disproportionation (and under what conditions) in aqueous solution:
(a) Fe2+;
(b) Sn2+;
(c) [ClO3]−
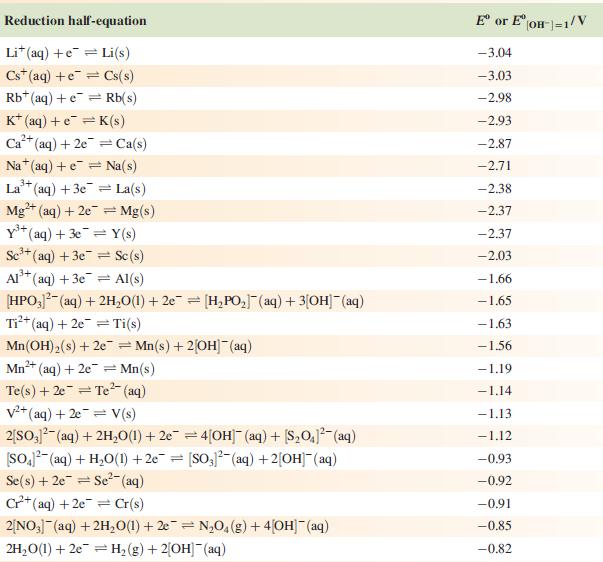
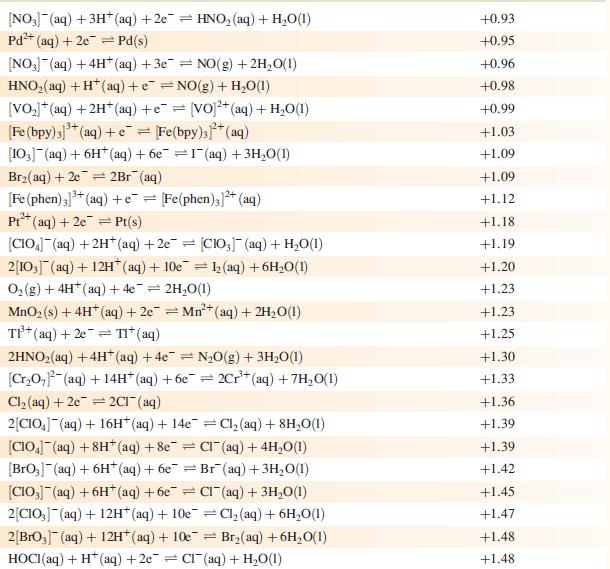
Data from Appendix 11
The concentration of each aqueous solution is 1 mol dm−3 and the pressure of a gaseous component is 1 bar (105 Pa). (Changing the standard pressure to 1 atm (101 300 Pa) makes no difference to the values of Eo at this level of accuracy.) Each half-cell listed contains the specified solution species at a concentration of 1 mol dm−3; where the half-cell contains [OH]−, the value of Eo refers to [OH−] = 1 mol dm−3, hence the notation Eo[OH−] = 1


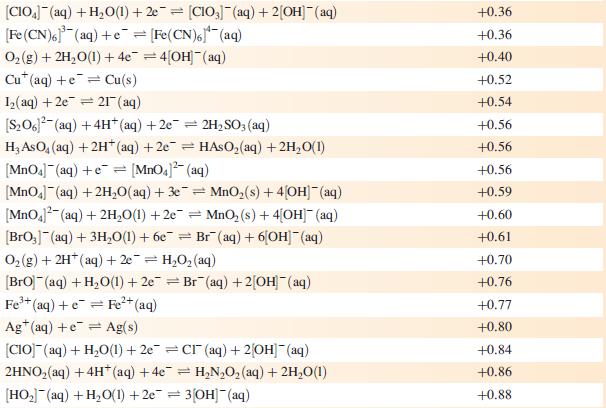

Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Question Posted:





