Using eq. 1.16, determine the energies of atomic orbitals of hydrogen with n = 1, 2, 3,
Question:
Using eq. 1.16, determine the energies of atomic orbitals of hydrogen with n = 1, 2, 3, 4 and 5. What can you say about the relative spacings of the energy levels?
Data from Equation 1.16
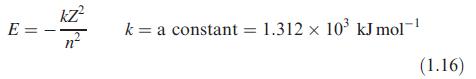
Transcribed Image Text:
E= kZ² nº k= = a constant = 1.312 x 10³ kJ mol-1 (1.16)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
Now we can use the given formula to determine the energies of the atomic orbitals of hydrogen with d...View the full answer

Answered By

Mary Boke
I have teached the student upto class 12th as well as my fellow mates.I have a good command in engineering,maths and science.I scored 90+ marks in 10th and 12th in maths.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The data shown in Table 6E.4 are the deviations from nominal diameter for holes drilled din a carbon-fiber composite material used in aerospace manufacturing. The values reported are deviations from...
-
The 32 teams in the National Football League (NFL) are worth, on average, $1.17 billion, 5% more than last year. The following data show the annual revenue ($ millions) and the estimated team value...
-
The Forest Service is deciding how to allocate its recreational funding. It can increase fishing opportunities in an area (for instance, by stocking), or it can build more hiking trails. It presents...
-
Grimm Company has 2,400,000 shares of common stock outstanding on December 31, 2014. An additional 150,000 shares of common stock were issued on July 1, 2015, and 300,000 more on October 1, 2015. On...
-
Purpose: To help you understand the importance of cash flows in the operation of a small business. You have just received your year-end financial statements from your CPA and you notice one very...
-
As of 2016, which of the following accurately represents the full FUTA rate and wage base? A. 0.6%, $118,500 B. Varies by state C. 6%, $7,000 D. 0.6%, $7,000
-
In 2014, political consulting firm Cambridge Analytica developed an app designed to create digital profiles of individuals via their information. Cambridge Analytica collected the data by inviting...
-
Gold Star Rice, Ltd., of Thailand exports Thai rice throughout Asia. The company grows three varieties of riceFragrant, White, and Loonzain. (The currency in Thailand is the baht. which is denoted by...
-
A manufacturing company reports the following information. Raw materials inventory, ending Raw materials used Current Year $ 169,500 2,160,000 1 Year Ago 2 Years Ago $ 190,500 2,522,000 $ 197,500...
-
How many radial nodes does each of the following orbitals possess: (a) 2s; (b) 4s; (c) 3p; (d) 5d; (e) 1s; (f ) 4p
-
Do the three 4p atomic orbitals possess the same or different values of (a) Principal quantum number, (b) The orbital quantum number and (c) The magnetic quantum number? Write down a set of quantum...
-
Find the equation of the tangent line to the parabola y2 = -18x that is parallel to the line 3x - 2y + 4 = 0.
-
Explain the rationale for the accounting treatment of compound financial instruments under IAS 32. Do you agree with IAS 32s stance?
-
During 2007, the U.S. economy was hit by a price shock when the price of oil increased from around $60 per barrel to around $130 per barrel by June 2008. While inflation increased during the fall of...
-
Scenario A A Co acquired a controlling interest in B Co and entered into the following transactions on acquisition date, 1 July 20x3. Required: Prepare the journal entries that A Co would record to...
-
Some Federal Reserve officials have discussed the possibility of increasing interest rates as a way of fighting potential increases in expected inflation. If the public came to expect higher...
-
On 1 July 20x3, Small Ltd issued 10 million new shares to owners of Sumo Pte Ltd in exchange for all of Sumo Pte Ltds shares. The share exchange is the result of a reverse acquisition of Small Ltd by...
-
Shamsud Ltd. operates on a calendar-year basis. At the beginning of December 2016, the company had the following current liabilities on its books: Accounts payable ........... $85,000 Rent payable...
-
Assessing simultaneous changes in CVP relationships Braun Corporation sells hammocks; variable costs are $75 each, and the hammocks are sold for $125 each. Braun incurs $240,000 of fixed operating...
-
Write equations to account for the acidity of CO 2 and boric acid (B(OH) 3 ), each dissolved in water.
-
Explain the following observations: (a) The heat of adduct formation of BEt 3 with NMe 3 is 72.3 kJ mol 1 , whereas that of B(OMe) 3 with the same amine is 31.5 kJ mol 1 . (b) The heat of adduct...
-
A standard procedure for improving the detection of the stoichiometric point in titrations of weak bases with strong acids is to use acetic acid as a solvent. Explain the basis of this approach.
-
A block of mass 1.3 kg is attached to a horizontal spring that has a force constant 1 200 N/m as shown in the figure below. The spring is compressed 2.0 cm and is then released from rest. a F x x = 0...
-
A 2kg block original at rest is being pushed by a force F at an angle of 0-30 below horizintal. The mass of the block is 2kg. The coefficients of static and kinetic friction between the block and the...
-
A small block of mass m slides in a vertical circular trajectory along the inside of a frictionless surface as shown. The magnitude of the normal force at point C is 6.8mg. The magnitude of the net...

Study smarter with the SolutionInn App


