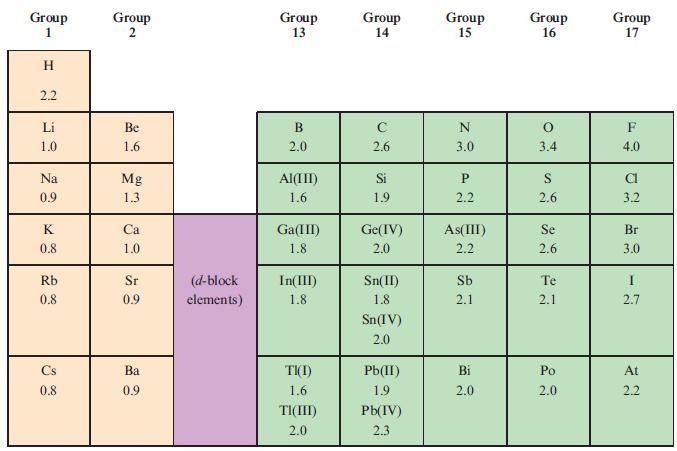
Using the data in Table 2.2, determine which of the following covalent single bonds is polar and
Question:
Using the data in Table 2.2, determine which of the following covalent single bonds is polar and (if appropriate) in which direction the dipole moment acts.
(a) N—H;
(b) F—Br;
(c) C—H;
(d) P—Cl;
(e) N—Br.
Table 2.2
Transcribed Image Text:
Group 1 I H 2.2 Li 1.0 Na 0.9 K 0.8 Rb 0.8 Cs 0.8 Group 2 Be 1.6 Mg 1.3 Ca 1.0 Sr 0.9 Ba 0.9 (d-block elements) Group 13 B 2.0 Al(III) 1.6 Ga(III) 1.8 In(III) 1.8 TI(I) 1.6 TI(III) 2.0 Group 14 с 2.6 Si 1.9 Ge(IV) 2.0 Sn(II) 1.8 Sn(IV) 2.0 Pb(II) 1.9 Pb(IV) 2.3 Group 15 N 3.0 P 2.2 As(III) 2.2 Sb 2.1 Bi 2.0 Group 16 O 3.4 S 2.6 Se 2.6 Te 2.1 Po 2.0 Group 17 F 4.0 CI 3.2 Br 3.0 I 2.7 At 2.2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
a NH Electronegativity difference 304 220 084 Since the electronegativity di...View the full answer

Answered By

Deepak Sharma
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Using the data in Table 116 on page 300, indicate the semiannual interest payment dates for the Motorola bonds that mature in 2031. (For the item in question, look under Interest Dates.) The two...
-
Using the data in Table 4-3, calculate the G° for ring flip to the other conformation of the molecules depicted in Problem 30. Make sure that the sign (i.e., positive or negative) of your values...
-
Using the data in the following table, predict the sign and magnitude of ÎH° for each of the following reactions. In each case, identify whether the reaction is expected to be endothermic...
-
The total cost of 2 chair and 1 table is 210 dollars. The total cost of 1 chair and 2 tables is 285 dollars. What is the cost of 1 chair?
-
Tucker Stevens opened an accounting firm on October 1, 2012. During the month of October, the business completed the following transactions: Oct 1 The business sold $50,000 of common stock to open...
-
Use the total wage bill rules and the labor demand schedule in Question 4 to determine whether demand is elastic or inelastic over the $6 to $11 wage rate range. Compute the elasticity coefficient...
-
After the positrons were annihilated, the energy density of the universe was dominated by the photons and the neutrinos. Show that the energy density in that era was given by \(u_{\text {total...
-
GB Service Corporation has two service departments, Administration and Accounting, and two operating departments, East and West. Administration costs are allocated on the basis of employees, and...
-
The current in the 1 3 . 8 2 resistor is / 3 = 0 . 9 A . 1 . Find the current in the other resistors in thecircuit? 2 . What is the total energy dissipated in resistor 4 1 7 . 2 0 if the power supply...
-
Use the Lewis structure model to deduce the type of nitrogennitrogen bond present in (a) N 2 H 4 , (b) N 2 F 4 , (c) N 2 F 2 (d) [N 2 H 5 ] + .
-
One member of each of the following sets of compounds is not isoelectronic with the others. Which one in each set is the odd one out? (a) [NO], CO, [NO] and [N3] (b) [CN], N, CO, [NO] and [0]- (c)...
-
The N 2 O 4 NO 2 reversible reaction is found to have the following equilibrium partial pressures at 100C. Calculate Ksp for the reaction. NO4(8) 0.0014 atm 11 2 NO(g) 0.092 atm
-
Describe the two primary objectives of macroeconomic stabilization policy.
-
Identify the four main categories of government spending and give an example of each. What are the governments four main revenue sources?
-
Although Okuns law holds for different countries, those with more flexible labor markets experience a higher response of unemployment to changes in GDP. During the recent financial crisis, real GDP...
-
What principal-agent problems resulted from the originate-to-distribute mortgage lending model?
-
Explain the processes of cost-push and demand-pull inflation. How do macroeconomists distinguish between the two?
-
Identify and discuss the criteria that are used to distinguish finance leases from operating leases.
-
A circular concrete shaft liner with Youngs modulus of 3.4 million psi, Poissons ratio of 0.25, unconfined compressive strength 3,500 psi and tensile strength 350 psi is loaded to the verge of...
-
Consult the Ellingham diagram in Fig. 6.16 and determine if there are any conditions under which aluminium might be expected to reduce MgO. Comment on these conditions. Figure 6.16. A,G*/ (kJ md-)...
-
In Fig. 6.11, which of the boundaries depend on the choice of Fe 2+ concentration as 10 5 mol dm 3 ? Figure 6.11. +0.8 +0.4 E/V -0.4 -0.8 Fe3+ Fe2+ H,O/H, 024 Fe(OH),(s) Fe(OH), (s) 6 8 10 12 14 pH
-
Adding NaOH to an aqueous solution containing Ni 2+ results in precipitation of Ni(OH) 2 . The standard potential for the Ni 2+ /Ni couple is 0.25 V and the solubility product K sp = [Ni 2+ ] [OH ]...
-
Identify 5 key questions that HR leaders should consider specific to the trend of Redefining Remote and Hybrid Work Strategies.
-
Assume that you are telling a STAR story. Roll over each item to read the complete sentence. Place each of the items into the most appropriate component of this story: situation, tasks, actions,...
-
Elaborate in 500 words on the advantages that administrative officers and secretaries can gain by pursuing a degree in library and information science. Additionally, discuss the significance of...

Study smarter with the SolutionInn App


