What hybridization scheme would be appropriate for the C atom in [CO3] 2 ? Draw resonance structures
Question:
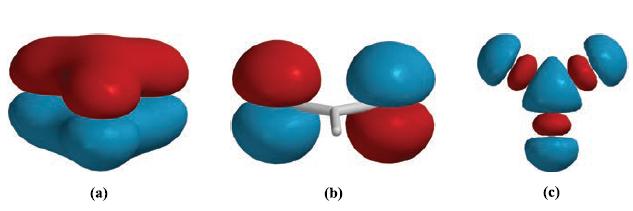
What hybridization scheme would be appropriate for the C atom in [CO3]2‾? Draw resonance structures to describe the bonding in [CO3]2‾. Figure 5.34 shows representations of three MOs of [CO3]2‾. The MOs in diagrams (a) and (b) in Fig. 5.34 are occupied; the MO in diagram (c) is unoccupied. Comment on the characters of these MOs and assign a symmetry label to each orbital.
Figure 5.34.
Transcribed Image Text:
(a) 88 (b) (c)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (5 reviews)
Hybridization scheme for the C atom in CO32 The C atom in CO32 is sp2 hybridizedThis is because it forms three sigma bonds with the three O atomsand t...View the full answer

Answered By

JAPHETH KOGEI
Hi there. I'm here to assist you to score the highest marks on your assignments and homework. My areas of specialisation are:
Auditing, Financial Accounting, Macroeconomics, Monetary-economics, Business-administration, Advanced-accounting, Corporate Finance, Professional-accounting-ethics, Corporate governance, Financial-risk-analysis, Financial-budgeting, Corporate-social-responsibility, Statistics, Business management, logic, Critical thinking,
So, I look forward to helping you solve your academic problem.
I enjoy teaching and tutoring university and high school students. During my free time, I also read books on motivation, leadership, comedy, emotional intelligence, critical thinking, nature, human nature, innovation, persuasion, performance, negotiations, goals, power, time management, wealth, debates, sales, and finance. Additionally, I am a panellist on an FM radio program on Sunday mornings where we discuss current affairs.
I travel three times a year either to the USA, Europe and around Africa.
As a university student in the USA, I enjoyed interacting with people from different cultures and ethnic groups. Together with friends, we travelled widely in the USA and in Europe (UK, France, Denmark, Germany, Turkey, etc).
So, I look forward to tutoring you. I believe that it will be exciting to meet them.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
(a) What hybridization scheme would be appropriate for the Si atom in SiH 4 ? (b) To which point group does SiH 4 belong? (c) Sketch a qualitative MO diagram for the formation of SiH 4 from Si and an...
-
What types of graphs would be appropriate for the following data? Circle as many types as are appropriate and would give clarity to the data. Use the following key: SL = stem-and-leaf H = histogram F...
-
What types of graphs would be appropriate for the following data? Circle as many types as are appropriate and would give clarity to the data. Use the following key: SL = stem-and-leaf H = histogram F...
-
Which of the following statements best describes the 'dilation' in rock mechanics? a. Movement of the rock along the shear direction during the direct shear tests b. Radial expansion of the intact...
-
After watching the video on outsourcing, discuss the following: 1. What role does the sourcing agent play in the 4Ps of marketing? 2. How does the outsourcing approach challenge the requirements of...
-
What seems to be wrong with the way the NPV of each project has been calculated? Indicate without any calculations, how Russ and Art should go about recalculating the projects NPVs.
-
Juliette Shulof Furs (JSF) was a New York corporation that had been in the fur-dealing business for 15 years. George Shulof, an officer of JSF, attended two auctions conducted by Finnish Fur Sales...
-
On August 1, 2010, Colombo Co.s treasurer signed a note promising to pay $240,000 on December 31, 2010. The proceeds of the note were $232,000. Required: a. Calculate the discount rate used by the...
-
Step 3: Using the Price and Total Quantity Demanded data from the table above, graph the demand curve on the axes provided (be sure to provide numbers on the axes for price and quantity demanded)....
-
Use a ligand group orbital approach to describe the bonding in [NH 4 ] + . Draw schematic representations of each of the bonding MOs.
-
In [B 2 H 7 ] (5.11), each B atom is approximately tetrahedral. (a) How many valence electrons are present in the anion? (b) Assume that each B atom is sp 3 hybridized. After localization of the...
-
Belle Equipment Co. wants to prepare interim financial statements for the first quarter of 2023. The company uses a periodic inventory system but would like to avoid making a physical count of...
-
Beta Corporation is making plans for the next fiscal year. Betas current sales are $3.5 million, expected to increase to $5 million, based on current assets of $2.4 million and fixed assets of $2.5...
-
Design another experiment using difference-indifferences to understand the effect of a policy change at your college.
-
The Daher Trucking Company needs to expand its fleet by 20 percent to meet the demands of two major contracts it just received to transport aeronautic equipment from manufacturing facilities...
-
Garr Company estimates its investment to be $0.25 in assets for each dollar of new sales. By each dollar of additional sales $0.04 profits will be produced and $0.01 can be reinvested in the company....
-
Grays Accounting pays Rita Flores $ 51,000 per year. Flores works 1,000 hours per year. Requirements 1. What is the hourly cost to Gray Accounting of employing Flores? Assume a 25-hour week and a...
-
If your instructor has assigned the Appendix to this chapter, redo Problem AP7-5 assuming that the company uses a periodic inventory system. In Problem AP4-5 The following information relates to...
-
In your readings, there were many examples given for nurturing close family relationships in this ever-evolving technological society we live in Based upon your readings and research describe three...
-
In their article Targeting and delivery of platinum-based anticancer drugs (Chem. Soc. Rev. 2012, 42, 202), X. Wang and Z. Guo review the expanding field of nanoparticle-based drug delivery....
-
Compounds of Au(III) are under investigation as anticancer drugs. Predict some of the similarities and contrasts with Pt(II) compounds.
-
Reductive dehalogenases are cobalamin-containing enzymes which catalyse the replacement of a halogen atom by a hydrogen atom in organic molecules (X is typically Cl). With reference to the principles...
-
Describe the Daily Construction Diary, In your description, include. 1. why this is such an important document and who should maintatain one? 2. some of the information that should be recorded each...
-
Find the APR in each of the following cases: You must show all your work to earn credits. APR Number of Times compounded per year EAR ? Semiannually 11.0% ? Monthly 11.9% ? Weekly 9.6%
-
Using the following table, identify the key benefits for each the 3 pest control strategies by outlining the risks associated with their respective use. and how to evaluate the results? strategy Risk...

Study smarter with the SolutionInn App


