Which of the following processes are expected to be exothermic? Give reasons for your answers. (a) Na
Question:
Which of the following processes are expected to be exothermic? Give reasons for your answers.
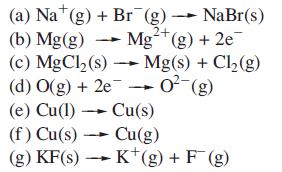
Transcribed Image Text:
(a) Na¹ (g) + Br (g) → NaBr(s) (b) Mg(g) Mg²+ (g) + 2e 2+ (c) MgCl₂ (s) →→→ Mg(s) + Cl₂(g) (d) O(g) + 2e 0² (g) (e) Cu(1)→ Cu(s) (f) Cu(s)→ Cu(g) (g) KF(s) → K+ (g) + F(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
Exothermic reactions release energy in the form of heat while endothermic reactions absorb energy a ...View the full answer

Answered By

Saleem Abbas
Have worked in academic writing for an a years as my part-time job.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
KYC's stock price can go up by 15 percent every year, or down by 10 percent. Both outcomes are equally likely. The risk free rate is 5 percent, and the current stock price of KYC is 100. (a) Price a...
-
Which of the following processes would you expect to be under control, and which would you expect not to be under control? Explain briefly why or why not. (a) Daily sales at each checkout line in a...
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
Compute the Regular Pay, Overtime Rate, Overtime Pay, and Gross Pay for each employee. Employees are paid weekly. Filing Employee Status Henry Smith Parker Liam Melanie Total S S S M S Hours Pay...
-
Using a resource-based view, explain why some firms improve their economic performance by adopting a CSR strategy, whereas others achieve either no results or damaging results.
-
A segment of human DNA has the following sequence of nucleotide pairs: When this segment of DNA is transcribed by RNA polymerase, what will be the sequence of nucleotides in the RNA transcript?...
-
Identify and describe the key attributes that might mitigate a red flag, suggesting that no bad act or compliance issues exist.
-
Thurgood Devices uses activity-based costing to allocate overhead costs to customer orders for pricing purposes. Many customer orders are won through competitive bidding. Direct material and direct...
-
Westeros, Inc. receives scabbards (sheath for a sword) from two different suppliers. 70 percent of its scabbards come from Knights Watch, LLC, while 30 percent come from Crow, Inc. Records indicate...
-
When nickel(II) oxide is heated in O 2 , some of the cations are oxidized and vacant cation sites are formed according to the equation: where h + denotes a vacant cation site and (s) denotes an ion...
-
Using data from the Appendices and the fact that calculate a value for the lattice energy of BaCl 2 . Outline any assumptions that you have made. AH(298K) = -859 kJ mol-,
-
Are male college students more easily bored than their female counterparts? This question was examined in the article Boredom in Young AdultsGender and Cultural Comparisons (J. Cross-Cult. Psych....
-
Explain the difference between (a) public offerings and private placements, (b) primary markets and secondary markets, (c) the money market and the capital market, and (d) organized security...
-
What is the dividend growth model, and how is it used to estimate the cost of common equity financing?
-
What are the three major functions of an investment banker?
-
Identify the primary drawbacks of a corporation.
-
Show the differences between organizational forms using the following criteria: (a) number of owners, (b) liability for firms debts, (c) change in ownership dissolves the firm, and (d) taxation.
-
What does it mean if management determines that one piece of its manufacturing equipment is impaired?
-
The words without recourse on an indorsement means the indorser is: a. not liable for any problems associated with the instrument. b. not liable if the instrument is dishonored. c. liable personally...
-
The structure of B 2 O 3 is formed from vertex linked BO 3 planar triangular units while in Al 2 O 3 aluminium ions adopt solely octahedral coordination to oxygen. Explain these observations on the...
-
11 B-NMR is an excellent spectroscopic tool for inferring the structures of boron compounds. With 11 B 11 B coupling ignored, it is possible to determine the number of attached H atoms by the...
-
Starting with B 10 H 14 and other reagents of your choice, give the equations for the synthesis of [Fe(nido-B 9 C 2 H 11 ) 2 ] 2 , and sketch the structure of this species.
-
Consider a 6% T-note with 1.5 years to maturity. Spot rates (expressed as semiannual yields to maturity) are: 6 months = 5%, 1 year = 6% and 1.5 years = 7%. If the note is selling for $992, compute...
-
Assume the firm invests $110,000 today to get $29,000 at Year 1 (i.e. one year from now), $26,000 at Year 2, $35,000 at Year 3, $43,000 at Year 4, $27,500 at Year 5, and $16,500 at Year 6. Assuming...
-
In the Single Index Model (SIM), how is the unsystematic risk of a security measured? Question 18Answer a. By the beta coefficient b. By the R-squared value c. By the residual return d. By the...

Study smarter with the SolutionInn App


