Consider the equilibrium between copper and its oxide: The Gibbs energy of formation of Cu2O is given
Question:
Consider the equilibrium between copper and its oxide: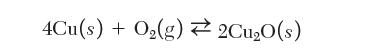
The Gibbs energy of formation of Cu2O is given by![]()
Make a plot of pO2 vs. T, illustrating where Cu is stable and where Cu2O is stable in the temperature range of 300 K to 1300 K.
Transcribed Image Text:
4Cu(s) + O(g) 2Cu0(s)
Step by Step Answer:

This question has not been answered yet.
You can Ask your question!
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
The equilibrium pressure of H, over solid uranium and uranium hydride, UH3' at 500 K is 139 Pa. Calculate the standard Gibbs energy of formation ofUH3 (s) at 500 K.
-
The Soviet Venera VII probe, which reached Venus on December 15, 1970, found the following conditions on the planet surface: Interferometric measurements indicate that there is only a 10 to 20 K...
-
The following data represents the number of Grand Jury indictments for Gloucester County, New Jersey for a sample of 11 weeks selected from July 2010 through June 2011 as reproduced from that...
-
Suppose that the Medicare rate of hospital reimbursement is reduced. Explain why the costs may not be shifted to other patients in the short run.
-
How might the concept of Crowd funding impact the traditional donor relationship development process? What implications might this have on the organization's fundraising ability in the long-run?
-
n = 9, p = 0.4, x 3 A binomial probability experiment is conducted with the given parameters. Compute the probability of x successes in the n independent trials of the experiment.
-
Comment on the statement that the essential purpose of the user interface is to enable the user and the system to exchange messages. Provide two examples.
-
How might a company make strategic use of countertrade schemes as a marketing weapon to generate export revenues? What are the risks associated with pursuing such a strategy?
-
Connectifi Solutions is a recently launched small company, providing WiFi installation services to businesses. You have been hired by Connectifi as an engineer. Your boss is seeking your input on the...
-
Consider the reaction of CrCl2 with H2 to form solid Cr as follows: At Answer the following questions: (a) From these data, estimate the enthalpy of reaction. (b) In an attempt to increase the extent...
-
Hydrogen cyanide can be manufactured by reaction of acetylene and nitrogen: Calculate the equilibrium composition at 800 K and 1 bar. CH + N 2HCN
-
We saw in this chapter that in Tompkins County, New York, older bridges were in worse condition than newer ones. Tompkins is a rural area. Is this relationship true in New York City as well? Here are...
-
What offense would a person who destroyed or damaged data in someones computer have committed? If a person electronically accessed anothers computer without permission, would this constitute a...
-
In 2006 in Hudson v. Michigan, Justice Scalia, writing for a sharply divided Supreme Court, concluded that when it comes to knock-and-announce violations, [r]esort to the massive remedy of...
-
Should the NCAA be exempt from the antitrust laws? Why or why not?
-
What provisions would you include in a model statute making arson a crime? Would you provide for separate degrees of the offense?
-
The Supreme Court has created a good-faith exception to the exclusionary rule where police rely on a search warrant that is later held to be invalid because the magistrate erred in finding probable...
-
Distinguished Budget Presentation Award. You are a governmental accountant for a large municipality, and you have recently been assigned to the Budget office and charged with building a better budget...
-
Chloroplasts are illuminated until the levels of the Calvin cycle intermediates reach a steady state. The light is then turned off. How does the level of RuBP vary after this point?
-
The total cross-sections for reactions between alkali metal atoms and halogen molecules are given in the table below (R.D. Levine and R.B. Bernstein, Molecular reaction dynamics, Clarendon Press,...
-
For the thermal decomposition Of F2O by the reaction 2 F2O (g) 2 Fl (g) + 0z (g),) Czarnowski and H) Schumacher (Chem. Phys. Lett. 17, 235 (1972)) have suggested the following mechanism: (a) Using...
-
One of the most historically significant studies of chemical reaction rates was that by M. Bodenstein (z. physik. Chem. 29,295 (1899)) of the gas-phase reaction 2 Hl(g) -t H2(g) + I2(g) and its...
-
Exercise 9.2: A simple acetylene-oxygen coflow nonpremixed flame shall be con- sidered. Flow 1 consists only of oxygen (O2), flow 2 only of acetylene (CH). 9.4 Exercises 139 (a) Determine the mixture...
-
For the planetary gear train shown: N2 = 20t, N4 = 40t, N5 = 18t, 6 = 36t, N7 = 154t. 3 is the arm (carrier) connecting planet gears N4 and N5. The ring gear angular velocity is 100 rpm ccw with an...
-
5-31 Saturated water vapor at 8 bar is generated in a natural gas boiler at a rate of 0.25 kg/s. This steam is used for process heating in an industrial facility and returns to the boiler at the same...

Study smarter with the SolutionInn App


