Hydrogen cyanide can be manufactured by reaction of acetylene and nitrogen: Calculate the equilibrium composition at 800
Question:
Hydrogen cyanide can be manufactured by reaction of acetylene and nitrogen: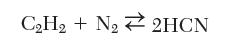
Calculate the equilibrium composition at 800 K and 1 bar.
Transcribed Image Text:
CH + N 2HCN
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
The following selected transactions were completed during the first year of operations: Jan. 3 Issued 15,000 shares of common stock at $23 per share for cash. 31 Issued 200 shares of common stock to...
-
Acetylene (C2H2) and nitrogen (N2) both contain a triple bond, but they differ greatly in their chemical properties. (a) Write the Lewis structures for the two substances. (b) By referring to...
-
Hydrogen cyanide, HCN, can be made by a two-step process. First, ammonia is reacted with O2 to give nitric oxide, NO. 4NH3(g) + 5O2(g) 4NO(g) + 6H2O(g) Then nitric oxide is reacted with methane,...
-
Discuss the ways that managed care organizations can infl uence the adoption of new technologies.
-
Which deal with advocacy and lobbying; governing and managing international and global organizations; and social entrepreneurship within the non-profit organization. Think of a non-profit...
-
An investor randomly purchases 12 stocks listed on a stock exchange. Historically, the probability that a stock listed on this exchange will increase in value over the course of a year is 50%. The...
-
What is consistency in user interface design? What problems might lack of consistency cause?
-
Research CaseGovernmental or Not-for-Profit Entity? In partnership with Jefferson County and the Mound City Visitors Bureau, Mound City recently established a Native American Heritage Center and...
-
Question 4 (8 Marks): For this question, your first task is to study the famous "Distance Vector (DV) Routing Algorithm" used at the network layer. You may refer to some slides I recently posted on...
-
Consider the equilibrium between copper and its oxide: The Gibbs energy of formation of Cu2O is given by Make a plot of pO2 vs. T, illustrating where Cu is stable and where Cu2O is stable in the...
-
Solve the multiple chemical reaction equilibrium problem in Example 9.19 at 800 K using the following set of independent reactions: Example 9.19 Consider a system initially charged with 1 mole of...
-
Under what conditions is travel time counted as time worked?
-
Should college athletes be paid? If not, why not? If so, how can one design a system that neither bankrupts most colleges nor discriminates against women?
-
Give some examples of actions that would probably fall within the conduct proscribed by (a) extortion and (b) vandalism or malicious mischief statutes.
-
What alternatives to the exclusionary rule might be adopted to enforce the protections of the Fourth Amendment? How effective are such alternatives likely to be?
-
If college sports are a public good, what can you say about the amount of it a university will supply? What can the universitys administration do to resolve this problem?
-
Should the offense of forgery be divided into degrees based on the importance of the forged documents? If so, what criteria would you propose for the various degrees of the crime?
-
The finance officer of a small city has heard that certain items of cost may be allowable under federal grants, even though they were not incurred specifically for the grant. To what source could the...
-
Show that gj concave AHUCQ Abadie For nonnegative variables, we have the following corollary.
-
(a) Distinguish between a step and a terrace. (b) Describe how steps and terraces can be formed by dislocations.
-
Distinguish between the following adsorption isotherms: Langmuir, BET, Temkin, and Freundlich.
-
Describe the essential features of the Langmuir-Hinshelwood, Eley-Rideal, and Mars van Krevelen mechanisms for surface-catalyzed reactions.
-
III. Derive the equations of motion of the shown system, then find the natural frequencies and mode vectors, with m = 20 kg, m = 40 kg, k = 1000 N/m, and k = 2000 N/m. Base k my 17(1) 00000 m 12(1)
-
1. A 2-D velocity field is given as (x,y) = Ay + Ax where A = 3 m/s/m, and coordinates are in meters a) Calculate the acceleration of a fluid particle at location (1,0) b) Determine the pressure...
-
Propane enters a well-insulated nozzle as a saturated vapor at 2 bar, with a velocity of 28 m/s, and exits at 1 bar with specific volume v=0.4439 m/kg. For steady-state operation, and neglecting...

Study smarter with the SolutionInn App


