Determine the second and third virial coeffi cients using the van der Waals equation of state. Begin
Question:
Determine the second and third virial coeffi cients using the van der Waals equation of state.
Begin by writing the van der Waals equation in compressibility factor form and performing a power-series expansion. The following mathematical relation is useful: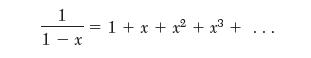
Transcribed Image Text:
1 1- x = 1+ x +x + x +
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)

Answered By

Aysha Ali
my name is ayesha ali. i have done my matriculation in science topics with a+ . then i got admission in the field of computer science and technology in punjab college, lahore. i have passed my final examination of college with a+ also. after that, i got admission in the biggest university of pakistan which is university of the punjab. i am studying business and information technology in my university. i always stand first in my class. i am very brilliant client. my experts always appreciate my work. my projects are very popular in my university because i always complete my work with extreme devotion. i have a great knowledge about all major science topics. science topics always remain my favorite topics. i am also a home expert. i teach many clients at my home ranging from pre-school level to university level. my clients always show excellent result. i am expert in writing essays, reports, speeches, researches and all type of projects. i also have a vast knowledge about business, marketing, cost accounting and finance. i am also expert in making presentations on powerpoint and microsoft word. if you need any sort of help in any topic, please dont hesitate to consult with me. i will provide you the best work at a very reasonable price. i am quality oriented and i have 5 year experience in the following field.
matriculation in science topics; inter in computer science; bachelors in business and information technology
_embed src=http://www.clocklink.com/clocks/0018-orange.swf?timezone=usa_albany& width=200 height=200 wmode=transparent type=application/x-shockwave-flash_
4.40+
11+ Reviews
14+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
In this problem we seek to develop an expression for the van der Waals constants a and b in terms of molecular parameters using the Sutherland model for potential energy. (a) Show that writing the...
-
Now assume that there are 1,500 identical firms in this competitive industry. That is, there are 1,500 firms, each of which has the cost data shown in the table. Complete the industry supply schedule...
-
The van der Waals equation of state, an approximate representation of the behavior of gases at high pressure, is given by Where a and b are constants having different values for different gases. (In...
-
Find the net torque on the wheel in the figure below about the axle through O perpendicular to the page, taking a = 7.00 cm and b = 25.0 cm. (Indicate the direction with the sign of your answer....
-
Explain what is meant by each of the following: (a) Freedom from error (b) Neutrality; (c) Completeness;
-
Prove the results in Eq. (8.7.53) in the chiral representation or any other representation that you care to use.
-
The race track is a fascinating example of financial market dynamics at work. Let's go to the track and make a wager. Suppose that, from a field of 10 horses, we simply want to pick a winner. In the...
-
Describe some important uses of electronic commerce and explain why it is important to accountants.
-
The Golf Academy books show that at the beginning of the year ( 1 / 1 / 2 0 1 6 , the beginning balance in accounts receivable was $ 8 , 0 0 0 . During the year the firm reported sales revenue of $ 4...
-
Determine the second and third virial coeffi cients using RedlichKwong equation of state. 1 1- x = 1 + x +x + 3 +
-
Consider a cylinder fi tted with a piston that contains 2 mol of H2O in a container at 1000 K. Calculate how much work is required to isothermally and reversibly compress this gas from 10 L to 1 L,...
-
What is Ella Company's current ratio? a. 0.69 b. 1.31 c. 3.88 d. 6.69 e. 2.39 Assets Cash ......... Accounts receivable..... Merchandise inventory.. Prepaid insurance Long-term investments.. Plant...
-
There are two parts to the presentation processpreapproach and approach. Distinguish between the two parts, and describe what occurs during each phase.
-
What personal characteristics does a salesperson need in order to develop and enhance a partnering relationship? Why is the conceptand perceptionof added value important in nurturing a relationship?
-
Describe the three main elements that comprise an effective presentation strategy. What principles should one consider adopting when developing a personal selling philosophy based on the...
-
Describe three predominant customer-oriented approaches for capturing the prospects attention and arousing interest.
-
Briefly describe the characteristics of an emotive style.
-
Barret Cameron, a student in Professor Wagners managerial accounting course, asked the following question. In the first accounting course, the teacher said inventory costs flow on a FIFO, LIFO, or...
-
If a test has high reliability. O the test measures what the authors of the test claim it measures O people who take the same test twice get approximately the same scores both times O scores on the...
-
The 18-electron rule is a general, but not universal, guide for assessing whether a certain transition-metal complex is stable or not. Both of the following are stable compounds, but only one obeys...
-
One of the main uses of the linear -olefins prepared by oligomerization of ethylene is in the preparation of linear low-density polyethylene. Linear low-density polyethylene is a copolymer produced...
-
Each of the following ethers has been shown to be or is suspected to be a mutagen, which means it can induce mutations in test cells. Write the structure of each of these ethers. (a) Chloromethyl...
-
Barney Draper (Draper) has been with the Surrey location for the past fifteen years, working in the warehouse. He started as a warehouse aid and was promoted to warehouse supervisor over the years....
-
Conduct research on existing/new 'Diversity Management' programs or initiatives in order that your team has a good understanding of how best to prepare your report/presentation. Discuss as a group...
-
Arguing in the affirmative. Topic: It is ethically appropriate for police and other government authorities to use Covid app data to help solve crimes. Identifies three major distinct arguments...

Study smarter with the SolutionInn App


