The following vaporliquid equilibrium data have been reported for a binary mixture of acetone (1) in chloroform
Question:
The following vapor–liquid equilibrium data have been reported for a binary mixture of acetone (1) in chloroform (2) at 35. 17°C. Test these data for thermodynamic consistency.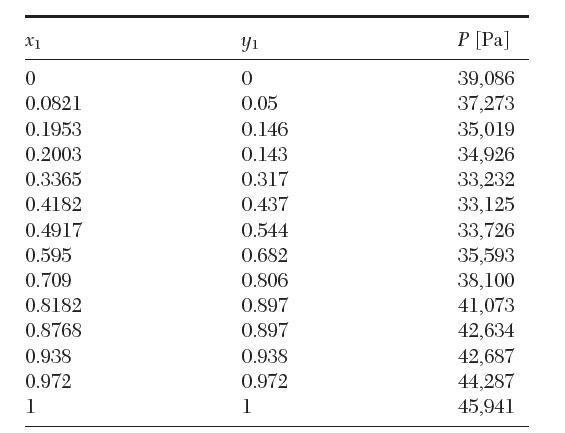
Transcribed Image Text:
X1 0 0.0821 0.1953 0.2003 0.3365 0.4182 0.4917 0.595 0.709 0.8182 0.8768 0.938 0.972 1 Y 0 0.05 0.146 0.143 0.317 0.437 0.544 0.682 0.806 0.897 0.897 0.938 0.972 1 P [Pa] 39,086 37,273 35,019 34,926 33,232 33,125 33,726 35,593 38,100 41,073 42,634 42,687 44,287 45,941
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)

Answered By

Susan Juma
I'm available and reachable 24/7. I have high experience in helping students with their assignments, proposals, and dissertations. Most importantly, I'm a professional accountant and I can handle all kinds of accounting and finance problems.
4.40+
15+ Reviews
45+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
Use ThermoSolver to fi nd the activity coeffi cient model parameters for the data presented in Problems 8.55 and 8.56. Problems 8.55 The following vaporliquid equilibrium data have been reported for...
-
The following vaporliquid equilibrium data have been reported for a binary mixture of acetone (1) in water (2) at 1 atm. Test these data for thermodynamic consistency. X1 0 0.015 0.036 0.074 0.175...
-
Your task in this problem will be to use a spreadsheet to generate a Txy diagram for a two-component system, using Raoult?s law to express the vapor?liquid equilibrium distribution of each species....
-
Perform the indicated operations and then simplify. Assume that all variables represent nonzero real numbers. 20yx3 + 15y4x + 25yx4 10yx
-
Two brothers, both of whom are certified public accountants (CPAs), form a professional association to provide tax-accounting services to the public. They also agree, in writing, that any disputes...
-
The functions f(x) = (x x ) x and g(x) = x (x x ) are different functions. For example, f(3) = 19,683 and g(3) 7.6 10 12 . Determine whether and are indeterminate forms and evaluate the limits. lim...
-
The fibers in a E-glass/epoxy composite are 0.0005 in. \((0.0127 \mathrm{~mm})\) in diameter before coating with an epoxy sizing \(0.0001 \mathrm{in} .(0.00254 \mathrm{~mm})\) thick. After the sizing...
-
In reviewing activity for July, the controller of Mathis, Inc., collected the following data concerning direct materials: Actual production . . . . . . . . . . . . . . . . . . . . . . . . . . . . . ....
-
what did you discover as you completed the holding on and letting go matrix ? In what ways could you minimize the positive reasons for holding on ? in what ways could you strengthen the advantages of...
-
You wish to fi t the benzene (1)isooctane (2) system to the following model for gE: The system temperature of interest is 200C. After a literature search, the only vaporliquid equilibrium data at...
-
Test the liquidvapor equilibrium data for the binary system of methanol (1)water (2) at 40C presented in Problem 8.53 for thermodynamic consistency by using the area test. Problem 8.53 Liquidvapor...
-
Suppose the only good you ever consume is Nestle's Crunch bars. What is your income elasticity of demand for Nestle's Crunch bars? What is your price elasticity of demand for Nestle's Crunch bars?
-
A research report produced by a dealer includes the following exhibit: Spot Rate Expected Spot Rate in One Year YEN/GBP 1.2317 1.2089 EUR/YEN 0.5367 0.5355 YEN/CHF 1.4267 1.4036 Calculate the spot...
-
Administration at the community hospital system (which includes an inpatient hospital, skilled nursing facility, durable equipment company, mental health facility, and outpatient counseling center)...
-
Before setting the new budget, what should we consider about view of the future obstacles? why
-
Question 1 Given recent legislation and the rising cost of gas heating, the use of heat pumps for heating homes during the winter season has increased. A heat pump with R134a heats a house by using...
-
What is the electric force from the rod on the point charge Q? Suppose that Q=13.5 nC and L = 3.41 cm. Break the rod up into four equal pieces and use superposition to find the force. X Z F = (...
-
What are the similarities and differences when conducting research in another country versus the domestic market?
-
PC Contractors, Inc., was an excavating business in Kansas City, Missouri. Union Bank made loans to PC, subject to a perfected security interest in its equipment and other assets, including...
-
Syntheses of each of the following compounds have been reported in the chemical literature. Using the indicated starting material and any necessary organic or inorganic reagents, describe short...
-
The following five-step synthesis has been reported in the chemical literature. Suggest reagents appropriate for each step.
-
Increased single-bond character in a carbonyl group is associated with a decreased carbonoxygen stretching frequency. Among the three compounds benzaldehyde, 2,4,6-trimethoxybenzaldehyde, and 2, 4,...
-
What is the difference between observed patterns of personality and a personality disorder? Although some patterns of behavior may contribute to an individual's personality, not all personality...
-
Review one of the clinical personality measures (i.e., Minnesota Multiphasic Personality Inventory-2, Millon Clinical Mutliaxial Inventory-III, Personality Assessment Inventory, Revised NEO...
-
the Jordan family has a health insurance policy with the following characteristics: a. Deductible: $1,000 b. Coinsurance Provision: 80%/20% c. Out of Pocket Maximum: $5,000 If total health expenses...

Study smarter with the SolutionInn App


