Two different compounds, both consisting of sodium (Na) and oxygen (O), were analyzed. The data are given
Question:
Two different compounds, both consisting of sodium (Na) and oxygen (O), were analyzed. The data are given below:
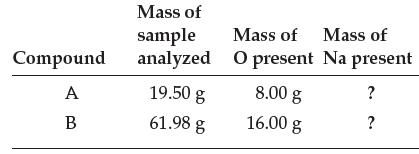
(a) Fill in the last column of the table.
(b) Calculate the %Na and %O for both compounds.
Transcribed Image Text:
Compound A B Mass of sample analyzed 19.50 g 61.98 g Mass of Mass of O present Na present ? ? 8.00 g 16.00 g
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)
To fill in the last column of the table we need to calculate the mass of sodium Na prese...View the full answer

Answered By

ANDREW KIPRUTO
Academic Writing Expert
I have over 7 years of research and application experience. I am trained and licensed to provide expertise in IT information, computer sciences related topics and other units like chemistry, Business, law, biology, biochemistry, and genetics. I'm a network and IT admin with +8 years of experience in all kind of environments.
I can help you in the following areas:
Networking
- Ethernet, Wireless Airmax and 802.11, fiber networks on GPON/GEPON and WDM
- Protocols and IP Services: VLANs, LACP, ACLs, VPNs, OSPF, BGP, RADIUS, PPPoE, DNS, Proxies, SNMP
- Vendors: MikroTik, Ubiquiti, Cisco, Juniper, HP, Dell, DrayTek, SMC, Zyxel, Furukawa Electric, and many more
- Monitoring Systems: PRTG, Zabbix, Whatsup Gold, TheDude, RRDtoo
Always available for new projects! Contact me for any inquiries
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
The following questions concern Poisson regression models fit to fictitious follow-up study data in which rates of disease are modeled as a function of age and smoking status. The SAS program codes...
-
Which of the following is an example of how managers use production cost reports to control costs? A. providing cost of goods sold for the income statement B. determining if newer, more efficient...
-
In 1950, J. R. Clarkson founded a family-owned industrial valve design and manufacturing company in Sparks, Nevada. For almost a half century, the company, known as the Clarkson Company, worked on...
-
1. What options does Personal Trainer have for developing a new system? What are some specific issues and options that Susan should consider in making a decision? 2. Susan has been asked to prepare a...
-
The following information was summarized from the balance sheets of the Coca-Cola Company and Subsidiaries at December 31, 2008, and PepsiCo Inc. and Subsidiaries at December 27, 2008: Required 1....
-
What is corporate governance, and how might it be improved for the benefit of the owners and customers of financial firms?
-
Hunter Corporation issued a \(\$ 100,000,61 / 2 \%, 10\)-year bond payable. Journalize the following transactions for Hunter. Include an explanation for each entry. a. Issuance of the bond payable at...
-
Foot Locker Inc. is the worlds number one retailer of athletic footwear and apparel. Headquartered in New York City, the company has over 44,000 employees and 3369 retail stores in 23 countries...
-
Marin Golf Inc. was formed on July 1, 2024, when Matt Magilke purchased the Old Master Golf Company. Old Master provides video golf instruction at kiosks in shopping malls. Magilke plans to integrate...
-
Bromine (Br) has two abundant isotopes, one with 44 neutrons and the other with 46 neutrons. Give the full atomic symbols for both isotopes.
-
An electromagnet bends the path of the fast moving ions in a mass spectrometer so they can reach the detector. The strength of an electromagnets magnetic field depends on the how much voltage is...
-
Consider the molecular electrostatic potential map for the BeH 2 molecule shown here. Is the hydrogen atom (shown as a white sphere) an electron acceptor or an electron donor in this molecule?
-
Indicate whether each of the following statements is true or false by writing T or F i n t he a nswer c olumn. There are numerous state, but no federal, regulations covering the misuse of computers...
-
What are income bonds and indexed bonds?
-
Lone Star Industries has just issued 160,000 of perpetual 10 per cent debt and used the proceeds to repurchase equity. The company expects to generate 75,000 of earnings before interest and taxes in...
-
Indicate whether each of the following statements is true or false by writing T or F i n t he a nswer c olumn. Employees who transfer money from their employers account into their own account without...
-
What is net operating working capital? Why does it exclude most short-term investments and notes payable?
-
Jill operates a sporting goods store in a rented location at a shopping mall. She is insured under a CGL policy with the following limits: General aggregate limit ............. $1,000,000...
-
Given the table below, about how much force does the rocket engine exert on the 4.0 kg payload? Distance traveled with rocket engine firing (m) Payload final velocity (m/s) 500 320 490 310 1020 450...
-
Identify whether each of the following compounds exhibits a molecular dipole moment. For compounds that do, indicate the direction of the net molecular dipole moment: a. CHCl 3 b. CH 3 OCH 3 c. NH 3...
-
Which of the following compounds has the larger dipole moment? Explain your choice: CHCl 3 or CBrCl 3
-
The specific rotation of ephedrine in ethanol (at 20C) is -6.3. A chemist prepared a mixture of ephedrine and its enantiomer, and this mixture had a specific rotation of -6.0. Calculate the % ee of...
-
1. What were three points mentioned by Chris that connected to the material we discussed this semester? 2. What was most interesting about this speaker? 3. Any other surprises or insights from the...
-
1. MS Excel, along with several other forms of spreadsheet software have proven to be very valuable tools for businesses of all kinds and sizes. Specifically, what are some key departments where...
-
CLO1: Consider the following method: public static int test(int n, int k) { if (k=0 || kn) { return 1; } else if (k > n) { return 0; } else { return test(n-1, k-1)+ test(n-1, k); } } For each of the...

Study smarter with the SolutionInn App


