Use the bottom graph on page 462 to determine what mass of water is required to dissolve
Question:
Use the bottom graph on page 462 to determine what mass of water is required to dissolve 50.0 g of NaNO3 at 40.0 °C.
Graph from Page 462
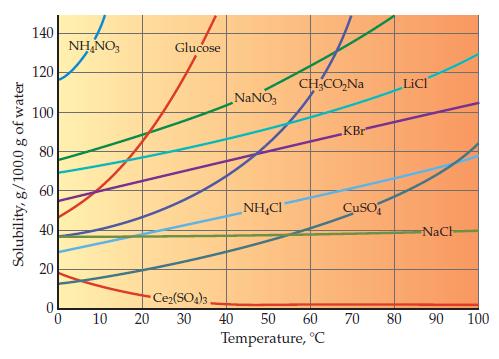
Transcribed Image Text:
Solubility, g/100.0 g of water 140 120 100 80 60 40 20 0 NH₂NO3 10 Glucose Ce₂(SO4)3. 20 30 NaNO3 NHẠC CH₂CO₂Na 40 50 60 Temperature, °C KBr CuSO LiCl NaCH 70 80 90 100
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
The provided graph displays solubility curves for various substances in water at diffe...View the full answer

Answered By

Shebla K
I am an MBA graduate having experience as an Assistant Professor at University level for two years. I always prepare well for a class as I believe that only if you become an ocean you can give a bucket of water. Being a teacher was not only my profession but also my passion.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
Examine the bottom graph on page 462 showing solubility in water as a function of temperature. What is the trend for most of the ionic substances shown? Data from Graph page 462 Solubility, g/100.0 g...
-
One ionic compound in the bottom graph on page 462 shows almost no temperature dependence, and one clearly violates the general trend. Identify these two ionic compounds. Data from Graph page 462...
-
(A) Calculate the quantity of that would be obtained if suggestions (1) and (2) in Example 14-4(b) were followed. Use data from Figure 14-10. What mass of water is needed to produce a saturated...
-
The following information has been extracted from the trial balance of M/s Randhir Transport Corporation. Adjustments 1. Closing stock for the year was Rs. 35,500. 2. Depreciation charged on plant...
-
Prepare the entries for the following transactions using a general journal: 1. Discarding an asset. (a) On January 4, shelving units, which had a cost of $7,200 and accumulated depreciation of...
-
The probability that equipment used in a hospital lab will need recalibration is given in the following table. A service firm is willing to provide maintenance and provide any necessary calibrations...
-
Parents of minors took Apple to court in 2012 for supplying game applications, on iPhones, that were free but through which users could purchase in-game currencies. Apparently, parents would log on...
-
On May 1, 2016, Benzs Sandwich Shop loaned $10,000 to Mark Henry for one year at 6 percent interest. Required Answer the following questions: a. What is Benzs interest income for 2016? b. What is...
-
Explain what a database management system is. Describe the functions of a database management system. 5 Paragraph Essay Intro ,Body , Conclusion
-
(a) How many grams of NaOH are needed to prepare 500.0 mL of a 0.300 M NaOH solution? (b) Describe how you would make this solution, including the equipment needed.
-
How many moles of potassium permanganate, KMnO 4 , are there in 28.86 mL of a 5.20 10 3 M solution of KMnO 4 ?
-
Figure 22.5 on page 744 shows growth rates in real GDP per hour worked in the United States for various periods from 1800 onward. How might the growth rates in the figure be different if they were...
-
If company A is sending money to Company B, then what is the disbursement float for Company A?
-
A spherical buoy of diameter 0.6m and mass 30kg is attached to the seabed by mooring rope and floats fully submerged. determine the tension in the mooring rope. the density of sea water is 1020kgm^-3.
-
5. For the 0-1 Knapsack Problem, the item #1 has weight of 4 and the value of $12; the item #2 has weight of 1 and the value of $1; the item #3 has weight of 5 and the value of $10; and the item #4...
-
Let = {0, 1} and consider the state-transition diagram given in Figure 1. 0 1 A 0 0 B D 1 0 Figure 1: State-transition diagram for Question 4. (a) Give examples of three strings that are accepted by...
-
What is the process involved in cleaving plasmid DNA using restriction enzymes, and how does this method contribute to molecular biology research and genetic engineering endeavors?
-
Suppose that 47% of all Americans have flown in an airplane at least once and that 28% of all Americans have ridden on a train at least once.What is the probability that a randomly selected American...
-
Consider the advantages and disadvantages of extending property rights so that everyone would have the right to prevent people imposing any costs on them whatsoever (or charging them to do so).
-
The sign is subjected to the uniform wind loading. Determine the stress components at points A and B on the 100-mm-diameter supporting post. Show the results on a volume element located at each of...
-
The 1-in.-diameter rod is subjected to the loads shown. Determine the state of stress at point B, and show the results on a differential volume element located at this point. 9 in. 200 lb B. 300 Ib...
-
The 1-in.-diameter rod is subjected to the loads shown. Determine the state of stress at point A, and show the results on a differential volume element located at this point. y 9 in. 200 Ib 300 Ib...
-
A taxpayer expenses out $ 1 5 0 , 0 0 0 of tangible property under IRC Section 1 7 9 when they file their Federal return. The taxpayer will need to apportion the Section 1 7 9 between California...
-
The purpose of this part is to provide you with a better understanding of the origins of the Codification. For this part, you should stay in the Codification, clicking on the tab Other Sources ( on...
-
A ticket is sold for a flat rate of $ 5 8 . 0 0 and the customer pays the $ 5 8 . 0 0 at the time of the sale. What will be the general journal entry for the company selling the ticket at a sales tax...

Study smarter with the SolutionInn App


