Use the electronegativity values in the chart on page 189 to calculate EN and predict whether the
Question:
Use the electronegativity values in the chart on page 189 to calculate ΔEN and predict whether the bonds (Ba—Cl, O—O, and Si—O) are covalent, polar covalent, or ionic.
O3
Data from Page 189
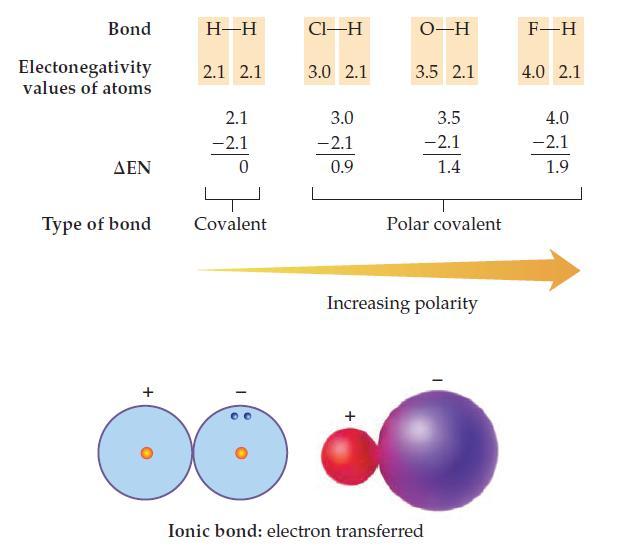
Transcribed Image Text:
Bond Electonegativity values of atoms ΔΕΝ Type of bond + H-H 2.1 2.1 2.1 -2.1 0 Covalent CI-H 3.0 2.1 3.0 -2.1 0.9 O-H 3.5 2.1 3.5 -2.1 1.4 Polar covalent Increasing polarity Ionic bond: electron transferred F-H 4.0 2.1 4.0 -2.1 1.9
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
Calculating EN BaCl Electronegativity of Ba 09 Electronegativity of Cl 30 EN 30 09 21 OO ...View the full answer

Answered By

Aysha Ali
my name is ayesha ali. i have done my matriculation in science topics with a+ . then i got admission in the field of computer science and technology in punjab college, lahore. i have passed my final examination of college with a+ also. after that, i got admission in the biggest university of pakistan which is university of the punjab. i am studying business and information technology in my university. i always stand first in my class. i am very brilliant client. my experts always appreciate my work. my projects are very popular in my university because i always complete my work with extreme devotion. i have a great knowledge about all major science topics. science topics always remain my favorite topics. i am also a home expert. i teach many clients at my home ranging from pre-school level to university level. my clients always show excellent result. i am expert in writing essays, reports, speeches, researches and all type of projects. i also have a vast knowledge about business, marketing, cost accounting and finance. i am also expert in making presentations on powerpoint and microsoft word. if you need any sort of help in any topic, please dont hesitate to consult with me. i will provide you the best work at a very reasonable price. i am quality oriented and i have 5 year experience in the following field.
matriculation in science topics; inter in computer science; bachelors in business and information technology
_embed src=http://www.clocklink.com/clocks/0018-orange.swf?timezone=usa_albany& width=200 height=200 wmode=transparent type=application/x-shockwave-flash_
4.40+
11+ Reviews
14+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
Use the electronegativity values in the chart on page 189 to calculate EN and predict whether the bonds (BaCl, OO, and SiO) are covalent, polar covalent, or ionic. SiO 2 Data from Page 189 Bond...
-
Use the electronegativity values in the chart on page 189 to calculate EN and predict whether the bonds (BaCl, OO, and SiO) are covalent, polar covalent, or ionic. BaCl 2 Data from Page 189 Bond...
-
Use the electronegativity values in Figure 1.14 to predict which of the indicated bonds in each of the following sets is more polar. Tell the direction of the polarity in each. (a) ClOCH 3 or ClOCl...
-
The mass-to-charge ratio for the positive ion F+ is 1.97 107 kg/C. Using the value of 1.602 1019 C for the charge on the ion, calculate the mass of the fluorine atom. (The mass of the electron is...
-
Due to an increase in recent employee layoffs because of economic conditions and the increased risk of workplace violence, as well as an increase in domestic restraining orders that several employees...
-
Merchant Company had the following foreign currency transactions: 1. On November 1, 20X6, Merchant sold goods to a company located in Munich, Germany. The receivable was to be settled in European...
-
In 2014, Barker contacted Price about a van Price had advertised for sale. The advertisement described the van as a 1994 Ford E350. Barker and Price agreed to meet, and, on April 9, Barker inspected...
-
Assume that Denis Savard Inc. has the following accounts at the end of the current year. 1. Common Stock. 2. Discount on Bonds Payable. 3. Treasury Stock (at cost). 4. Notes Payable, short-term. 5....
-
Discuss the business benefits of adopting the relational database model within an organization. How does a relational model create or enhance business value? Are there any limitations to using a...
-
Without knowing electronegativity values, a student claims BaCl 2 is more ionic than BeCl 2 . All she has access to is a periodic table. How does she know she is right?
-
Draw a dot diagram for NO + .
-
(a) Make recommendations to Harry and Belinda regarding where to seek financing and what APR to expect. (b) Using the Garman/Forgue companion website or the information in Table 7-1, calculate the...
-
While negotiating a possible buyout with the management of a firm, the CEO of the target starts to play hardball. He continues to add contingencies to the deal. In addition, he has recently raised...
-
You have seen many reports in the press about Adidas or Nike and how the working conditions of the foreign partners and suppliers are sometimes abysmal. Is this just a cost of doing business through...
-
As discussed in the text, in the absence of market imperfections and tax effects, we would expect the share price to decline by the amount of the dividend payment when the equity goes ex dividend....
-
Increasingly, firms such as P&G, Corning (www.corning. com) and Millennium Pharmaceuticals (www.millennium. com) claim to have a core competency and competitive advantage based on their ability to...
-
You are the cofounder and president of a new venture, manufacturing products for the recreational market. Five months after launching the business, one of your key suppliers informs you it can no...
-
Textron Manufacturing Inc. assembles industrial testing instruments in two departments, assembly and testing. Operating data for 2009 and 2010 follow: 2010 2009 Assembly department Actual direct...
-
Havel says the grocer doesnt believe what is on the sign and indeed, he says the grocers customers will barely notice it. But Havel maintains that the sign serves a specific function. How would you...
-
For IF ( = 610 cm 1 ) calculate the vibrational partition function and populations in the first three vibrational energy levels for T = 300 and 3000 K. Repeat this calculation for IBr ( = 269 cm 1 )....
-
Evaluate the vibrational partition function for H 2 O at 2000 K, where the vibrational frequencies are 1615, 3694, and 3802 cm 1 .
-
Evaluate the vibrational partition function for SO 2 at 298 K, where the vibrational frequencies are 519, 1151, and 1361cm 1 .
-
igs in the Is it ethical for a business owner who controls the business to escape liability for the business's contracts and torts by hiding behind the veil of the business organization? Is it...
-
Write a brief description of loan management and credit analysis and discuss the background information associated with the problems that need to be solved. Identify project goals and the underlying...
-
From A&D High Tech (B) Managing Scope Change What is the... From A&D High Tech (B) Managing Scope Change What is the projected completion date What is the total cost? How much of the total costs are...

Study smarter with the SolutionInn App


