Use the electronegativity values in the chart on page 189 to calculate EN and predict whether the
Question:
Use the electronegativity values in the chart on page 189 to calculate ΔEN and predict whether the bonds (Ba—Cl, O—O, and Si—O) are covalent, polar covalent, or ionic.
BaCl2
Data from Page 189
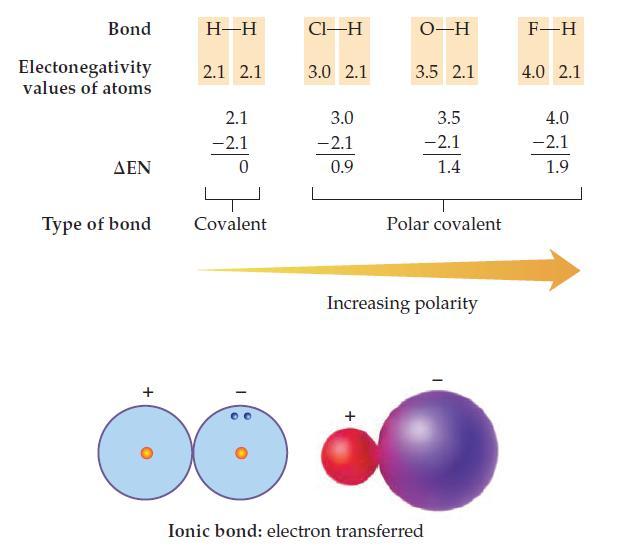
Transcribed Image Text:
Bond Electonegativity values of atoms ΔΕΝ Type of bond + H-H 2.1 2.1 2.1 -2.1 0 Covalent CI-H 3.0 2.1 3.0 -2.1 0.9 O-H 3.5 2.1 3.5 -2.1 1.4 Polar covalent Increasing polarity Ionic bond: electron transferred F-H 4.0 2.1 4.0 -2.1 1.9
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
EN values Ba 09 Cl 30 EN 21 The bond betw...View the full answer

Answered By

Fahmin Arakkal
Tutoring and Contributing expert question and answers to teachers and students.
Primarily oversees the Heat and Mass Transfer contents presented on websites and blogs.
Responsible for Creating, Editing, Updating all contents related Chemical Engineering in
latex language
4.40+
8+ Reviews
22+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
Use the electronegativity values in the chart on page 189 to calculate EN and predict whether the bonds (BaCl, OO, and SiO) are covalent, polar covalent, or ionic. SiO 2 Data from Page 189 Bond...
-
Use the electronegativity values in the chart on page 189 to calculate EN and predict whether the bonds (BaCl, OO, and SiO) are covalent, polar covalent, or ionic. O 3 Data from Page 189 Bond...
-
Use the electronegativity values in Figure 1.14 to predict which of the indicated bonds in each of the following sets is more polar. Tell the direction of the polarity in each. (a) ClOCH 3 or ClOCl...
-
Why does Erasmus attack Church officials in his In Praise of Folly? O For spending money on lavish art O For not allowing clergy to marry O For supporting military campaigns O For selling pardons and...
-
What should be included in disaster planning for a big employer in New York City that is concerned about terrorism attacks that might shut down the company and part of the city?
-
Harris Inc. had the following transactions: 1. On May 1, Harris purchased parts from a Japanese company for a U.S. dollar equivalent value of $8,400 to be paid on June 20. The exchange rates were May...
-
Heritage, a general contractor, had filed a breach of contract action against an electrical subcontracting firm that had withdrawn its bid right before the contract was to be performed. A jury had...
-
Union Express has 60 tons of cargo that needs to be shipped from Boston to Dallas. The shipping capacity on each of the routes Union Express planes fly each night is shown in the following table:...
-
Make a Relational Model Table and Define Keys from Entity Relation Diagram Q2: Design the World Music Association Database using given ERD. ORCHESTRA "Orchestra Name City Country Music Director...
-
Without knowing electronegativity values, a student claims BaCl 2 is more ionic than BeCl 2 . All she has access to is a periodic table. How does she know she is right?
-
Draw a dot diagram for NO + .
-
Indicate whether each of the following statements is true or false by writing T or F in t he a nswer c olumn. Touting occurs when an investor who owns shares of a companys stock posts notices online...
-
Your small manufacturing company is in serious financial difficulty. A large order of your products is ready to be delivered to a key customer, when you discover that the product is simply not right....
-
Refer to the box entitled How Would You Do That? 1. Pick another industry that is of interest to you. What did you identify as your indicator of potential market size? What market performance...
-
As you learned in the section exploring CAGE, the Foreign Corrupt Practices Act is a U.S. federal law that makes it illegal for a citizen or corporation of the United States or a person or...
-
Entrepreneurship starts with an idea. Without being critical or judgmental, brainstorm a set of 10 ideas that could lead to the startup of a new business. Screen your ideas and select those that...
-
Identify a company that has recently announced an acquisition. Study the terms of the deal and identify to the extent possible the market value of the target, its intrinsic value, and the acquisition...
-
Varceles Design has decided to experiment with two alternative manufacturing approaches, identified as MF and LI, for producing mens fashions. The firm expects the total demand to be 20,000 suits....
-
Representative data read from a plot that appeared in the paper Effect of Cattle Treading on Erosion from Hill Pasture: Modeling Concepts and Analysis of Rainfall Simulator Data (Australian Journal...
-
Evaluate the vibrational partition function for NH 3 at 1000. K for which the vibrational frequencies are 950., 1627.5 (doubly degenerate), 3335, and 3414 cm 1 (doubly degenerate). Are there any...
-
Evaluate the vibrational partition function for CFCl 3 at 298 K, where the vibrational frequencies are (with degeneracy in parentheses) 1081, 847 (2), 535, 394 (2), 350., and 241(2) cm 1 .
-
Determine the populations in n = 0 and 1 for H 81 Br ( = 2649 cm 1 ) at 298 K.
-
Kyle Corporation is comparing two different capital structures, an all-equity plan (Plan I) and a levered plan (Plan II). Under Plan I, the company would have 745,000 shares of stock outstanding....
-
Find the periodic payment R required to amortize a loan of P dollars over t years with interest charged at the rate of r %/year compounded m times a year. (Round your answer to the nearest cent.) P =...
-
You have $5,100 to deposit. Regency Bank offers 12 percent per year compounded monthly (1.0 percent per month), while King Bank offers 12 percent but will only compound annually. How much will your...

Study smarter with the SolutionInn App


