Use the electronegativity values in the chart on page 189 to calculate EN and predict whether the
Question:
Use the electronegativity values in the chart on page 189 to calculate ΔEN and predict whether the bonds (Ba—Cl, O—O, and Si—O) are covalent, polar covalent, or ionic.
SiO2
Data from Page 189
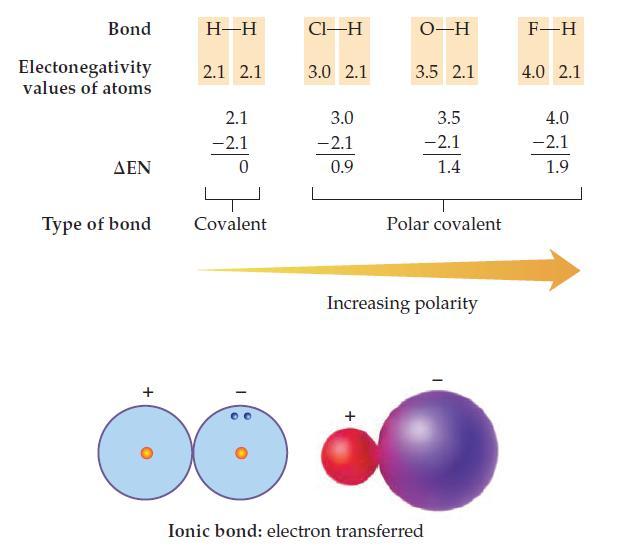
Transcribed Image Text:
Bond Electonegativity values of atoms ΔΕΝ Type of bond + H-H 2.1 2.1 2.1 -2.1 0 Covalent CI-H 3.0 2.1 3.0 -2.1 0.9 O-H 3.5 2.1 3.5 -2.1 1.4 Polar covalent Increasing polarity Ionic bond: electron transferred F-H 4.0 2.1 4.0 -2.1 1.9
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
To calculate EN and predict the type of bond in SiO2 we will use the same steps as before Calculatin...View the full answer

Answered By

Allan Simiyu
I am an adroit Writer. I am a dedicated writer having worked as a writer for 3 years now. With this, I am sure to ace in the field by helping students break down abstract concepts into simpler ideas.
5.00+
8+ Reviews
54+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
Use the electronegativity values in the chart on page 189 to calculate EN and predict whether the bonds (BaCl, OO, and SiO) are covalent, polar covalent, or ionic. O 3 Data from Page 189 Bond...
-
Use the electronegativity values in the chart on page 189 to calculate EN and predict whether the bonds (BaCl, OO, and SiO) are covalent, polar covalent, or ionic. BaCl 2 Data from Page 189 Bond...
-
Use the electronegativity values in Figure 1.14 to predict which of the indicated bonds in each of the following sets is more polar. Tell the direction of the polarity in each. (a) ClOCH 3 or ClOCl...
-
Determine the magnitude of the projection of the moment cause by the force about the a a axis. OKAY, SO I DONT UNDERSTAND HOW TO GET "R" BECAUSE THE FORCE IS MEASURED ON AN AXIS AND NOT A POINT OR...
-
Due to an increase in recent employee layoffs because of economic conditions and the increased risk of workplace violence, as well as an increase in domestic restraining orders that several employees...
-
The Hi-Stakes Company has a number of importing and exporting transactions. Importing activities result in payables and exporting activities result in receivables. (LCU represents the local currency...
-
Oncology Tech is a manufacturer of cancer therapy devices called compensators. CMS was a company that sold cancer treatment planning software for use in purchasing cancer treatment devices. In 2006,...
-
Special Revenue Fund, Voluntary Nonexchange Transactions. The City of Eldon applied for a competitive grant from the state government for park improvements such as upgrading hiking trails and bike...
-
. Do policies exist within the Linux workstation or server environment? If so, what are they? . Are there ways that Windows policies can be leveraged to enhance corporate or security policy?
-
Without knowing electronegativity values, a student claims BaCl 2 is more ionic than BeCl 2 . All she has access to is a periodic table. How does she know she is right?
-
Draw a dot diagram for NO + .
-
Determine the resultant moment produced by the forces about point O. 0.25 m 0.125 m, 0 0.3 m- 60 F = 500 N F = 600 N
-
Your company acquires another company in China. When you start to transform the organizational structure to that commonly used by your company at home and in other countries, you encounter...
-
Based on the information in the box entitled How Would You Do That? 1, assume that you have determined that established leaders have such an experience advantage that youll never catch their cost...
-
Review the opening vignette about the three bicycle manufacturers. Use the strategy-diamond and the generic strategy model to describe the positioning strategy of each firm. Based on what you know...
-
How Would You Do That? 2 applies a portfolio evaluation tool to Disney. Internally,Disney executives view one of their dynamic capabilities as that of being the best at creating world-class...
-
Try to apply the profit pool tool to another industry. Where would you turn for data to do this? How friendly is that data for the purposes of using this tool? If you are having trouble being...
-
Lawn Master manufactures riding lawn mowers that it sells to the large discount stores such as Wal-Mart, Lowes, and Home Depot. The mowers are marketed as a "value" product, with good quality at a...
-
Consider a game of poker being played with a standard 52-card deck (four suits, each of which has 13 different denominations of cards). At a certain point in the game, six cards have been exposed. Of...
-
When 4 He is cooled below 2.17 K it becomes a superfluid with unique properties such as a viscosity approaching zero. One way to learn about the superfluid environment is to measure the...
-
Calculate the vibrational partition function for H 35 Cl ( = 2990 cm 1 ) at 300 and 3000. K. What fraction of molecules will be in the ground vibrational state at these temperatures?
-
Determine the rotational partition function for I 35 Cl (B = 0.114 cm 1 ) at 298 K.
-
Fulkerson Manufacturing wishes to maintain a sustainable growth rate of 9.25 percent a year, a debtequity ratio of .40, and a dividend payout ratio of 32.5 percent. The ratio of total assets to sales...
-
Communication Plan Template Complete the Communication Plan Template. Identify all the stakeholders and resources that are noted within the case. Identify a minimum of two additional stakeholders or...
-
Saved You have $10,000 to invest in a stock portfolio. Your choices are Stock X with an expected return of 12.1 percent and Stock Y with an expected return of 9.8 percent. If your goal is to create a...

Study smarter with the SolutionInn App


