Using lines, solid wedges, and dashed wedges, draw the three-dimensional shape of diethyl ether, C 2 H
Question:
Using lines, solid wedges, and dashed wedges, draw the three-dimensional shape of diethyl ether, C2H6O. Indicate the numeric value of all bond angles.
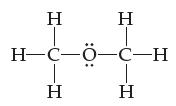
Transcribed Image Text:
Η H¬C- Η Η -C-H Η
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
H H H 109 109 1090 H 109 HO 1...View the full answer

Answered By

Shebla K
I am an MBA graduate having experience as an Assistant Professor at University level for two years. I always prepare well for a class as I believe that only if you become an ocean you can give a bucket of water. Being a teacher was not only my profession but also my passion.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
Using lines, solid wedges, and dashed wedges, draw the three dimensional shape of ethane, C 2 H 6 . Indicate the numeric value of all bond angles. H=C=C- -
-
The connections in CH 3 NO 2 are: (a) Put in the remaining valence electrons to complete the Lewis dot diagram. (b) Using lines, solid wedges, and dashed wedges, draw the three-dimensional shape of...
-
The connections in C 2 N 2 H 2 O are: (a) Put in the remaining valence electrons to complete the Lewis dot diagram. (b) Using lines, solid wedges, and dashed wedges, draw the three-dimensional shape...
-
An employee received the following benefits: regular compensation income, net of P25,000 SSS, PHIC & HDMF employee share - P850,000; 13th month pay - P120,000; De minimis benefits within their limits...
-
Mercury Athletics manufactures sporting goods that are then sold to retailers. It is a very competitive industry where quality and price are important to gain space on retailers shelves. Mercurys...
-
Astro Investment Bank has the following bond deals under way: Determine the net proceeds of each bond and the cost of the bonds for each company in terms of yield. The bond yield in the table is the...
-
1. Your instructor will divide the class into teams and assign each team the task of investigating the start-up of one of the following businesses: a. Submarine sandwich shop b. Day care service c....
-
Periodic versus Perpetual Entries Chippewas Company sells one product. Presented below is information for January for Chippewas Company. Jan. 1 Inventory 100 units at $6 each 4 Sale 80 units at $8...
-
Q6. Draw parallel realization of IIR by using the transfer function given by H(z) 3 (2 z+5Z+4)/ (2z+1) (z+2)
-
Using lines, solid wedges, and dashed wedges, draw the three-dimensional shape of the hydronium ion, H 3 O + . Indicate the numeric value of all bond angles. [H-Q-H]+ -C H
-
Using lines, solid wedges, and dashed wedges, draw the three-dimensional shape of acetaldehyde, C 2 H 4 O. Indicate the numeric value of all bond angles. O H || T H-C-C-H H
-
The financial statements of Apple Inc. are presented in Appendix A at the end of this textbook. Instructions for accessing and using the companys complete annual report, including the notes to the...
-
What is the gain/(loss) to be recognized in the income statement for 20x4 under IFRS 9? (a) $800,000 (b) 0 (c) $(100,000) (d) $(200,000) (e) $100,000 On 1 October 20x4, SingCo committed to purchase...
-
On 30 September 20x5, Singco entered into a non-cancellable contract to purchase inventory for 100,000 euros to be delivered on 31 January 20x6 with payment due on 31 March 20x6. Singco was concerned...
-
Alfalfa Companys functional currency is the dollar. On 30 June 20x1, it entered into a forward exchange contract to purchase FC 100,000 at the forward rate of $1.077 for delivery on 30 June 20x2. The...
-
The functional currency of K Co is the US dollar. On 1 January 2010, the management of K Co approved a decision to buy equipment for S$1,400,000. The equipment does not meet the conditions of a...
-
A Co has as its functional currency the Singapore dollar (S$) and enters into the following transaction. The business model of A Co is to hold investments to collect contractual cash flows and for...
-
Assume Neil, Inc., is expanding into Germany. The company must decide where to locate and how to finance the expansion. Identify the financial statement where these decision makers can find the...
-
The vapor pressure of the liquid NH, is measured at different temperatures. The following vapor pressure data are obtained. Temperature, K P, mmHg 217.1 223.4 234.7 588.1 Calculate the enthalpy of...
-
The product of an aldol condensation is an α,β unsaturated ketone which is capable of undergoing hydrogenation to yield a saturated ketone. Using this technique, identify...
-
Identify the reagents you would use to convert cyclohexanone into each of the following compounds. (a) (b) (c) (d) (e) (f) (g)
-
The enolate of a ketone can be treated with an ester to give a diketone. Draw a mechanism for this Claisen-like reaction, and explain why an acid source is required after the reaction is complete. 1)...
-
How do emerging technologies, such as super-resolution microscopy, single-cell imaging, and organelle proteomics, contribute to our understanding of organelle structure, function, and dynamics at a...
-
Researchers often obtain federal grants to support their projects. Some of the grant goes to various administrative offices at the University in what is/are known as ?
-
Entered a 6 year loan on 1 march current year for the purchase of equipment that was required for income producing purpose. The borrowing cost assosiated with loan were. 750. What is deductible...

Study smarter with the SolutionInn App


