Using lines, solid wedges, and dashed wedges, draw the three-dimensional shape of acetaldehyde, C 2 H 4
Question:
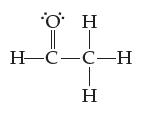
Using lines, solid wedges, and dashed wedges, draw the three-dimensional shape of acetaldehyde, C2H4O. Indicate the numeric value of all bond angles.
Transcribed Image Text:
O H || T H-C-C-H H
Step by Step Answer:

This question has not been answered yet.
You can Ask your question!
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
Using lines, solid wedges, and dashed wedges, draw the three dimensional shape of ethane, C 2 H 6 . Indicate the numeric value of all bond angles. H=C=C- -
-
The connections in CH 3 NO 2 are: (a) Put in the remaining valence electrons to complete the Lewis dot diagram. (b) Using lines, solid wedges, and dashed wedges, draw the three-dimensional shape of...
-
The connections in C 2 N 2 H 2 O are: (a) Put in the remaining valence electrons to complete the Lewis dot diagram. (b) Using lines, solid wedges, and dashed wedges, draw the three-dimensional shape...
-
Forward exchange contract designated as a fair value hedge of a foreign-currency-denominated accounts receivable, weakening $US On October 15, 2018, our company sells to a retailer located in Austria...
-
Comparing the results of cost allocations, using simplified costing versus activity-based costing Required: Referring to P9-6, compare the results of the cost allocations to the Young Products and...
-
Lunar Vacations needs to raise $6,000,000 for its new project (a golf course on the moon). Astro Investment Bank will sell the bond for a commission of 2.5%. The market is currently yielding 7.5% on...
-
1. Identify an entrepreneur in your area you would like to interview. 2. Contact the person you have selected and make an appointment. Be sure to explain why you want the appointment and to give a...
-
The comparative balance sheet of Wedge Industries Inc. for December 31, 2014 and 2013 is as follows: The following additional information is taken from the records: 1. Land was sold for $100. 2....
-
8. The electronic health record shows an order for 120 mL of 25 % formula to be administered in the nasogastric feeding tube every 2 hours between 0700 and 1900 each day. The 30 mL can of...
-
Using lines, solid wedges, and dashed wedges, draw the three-dimensional shape of diethyl ether, C 2 H 6 O. Indicate the numeric value of all bond angles. HC- -C-H
-
An atom has no lone pairs of electrons on it and four other atoms bound to it. Why is 109.5 the bond angle adopted by this molecule?
-
(a) Give two examples of applications of quantum wells. (b) Describe why quantum wells are used and if either molecular materials or traditional solid-state materials can exhibit similar properties....
-
Prism Co, a magazine publisher, reported net profit before tax of $1,300,000 for the year ended 31 December 20x1. The only disallowed expenses were the depreciation on private motor vehicles of...
-
The Heritage Index, published yearly by the Heritage Foundation, provides a comprehensive numerical measure of overall economic freedom for countries, with specific indicators reflecting the overall...
-
The capital structure of Model Company on 31 December 20x2 is as follows: The preference shares were convertible into ordinary shares in the ratio of 1,000 preference shares for 500 ordinary shares....
-
Improved transportation and storage technologies has made it easier for people in the United States to source their fresh fruit from abroad, and the majority of fruit is now imported, with much it...
-
How may an investor use the information on deferred taxes in financial analysis of an entity?
-
Answer these questions about two companies. 1. Emerald, Inc., began the year with total liabilities of $80,000 and total stockholders equity of $80,000. During the year, total assets increased by...
-
Fred Farmer needs to prepare a balance sheet for his bank. He spent the day getting the following information. Fred needs your help to build a balance sheet and evaluate it. The information was...
-
Beta-keto esters can be prepared by treating the enolate of a ketone with diethyl carbonate. Draw a plausible mechanism for this reaction. OEt 1) LDA 2) Eto OEt 3) ,*
-
The enolate of an ester can be treated with a ketone to give a b-hydroxy ester. Draw a mechanism for this aldol- like reaction. 1) LDA Eto Eto 2) 3) H,O*
-
Nitriles undergo alkylation at the α position much like ketones undergo alkylation at the α position. The α position of the nitrile is first deprotonated to...
-
Includes a complete discussion of income from student tuition compared to the overall outlay of salary for instructors and what other variables should be considered when looking at funding for...
-
When the "market" act as a control mechanism, an investor can buy large number of shares to become a major shareholder and influence the decision makings of the CEO and the firm. What is this...
-
If Bryan held the bonds until 21st November 2024, what was her holding period yield if the yield was 6.75%p.a.? on the day he sold the bond.

Study smarter with the SolutionInn App


