Provide the reagents needed to achieve each of the following transformations. (a) (b) (c) -OH Br
Question:
Provide the reagents needed to achieve each of the following transformations.
(a)
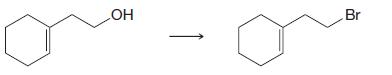
(b)
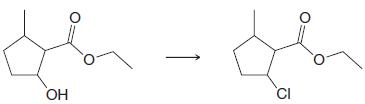
(c)

Transcribed Image Text:
-OH Br
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
In 1st reaction OH convert into CL by useing different types of regent such as HCL PCl 2 phosphor...View the full answer

Answered By

Shazni Ahamed
my self sudhanshu pandey , i have two year teaching experience . while teaching i have learned how to teach intresting way so that student can understand easily and they can retain for long time.And i can easily understand student problem
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry
ISBN: 978-1118875766
12th Edition
Authors: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Question Posted:
Students also viewed these Sciences questions
-
Provide the reagents needed to accomplish each of the following transformations. (a) (b) (c) (d) (e) (f) (g) (h) HO HO HO
-
Provide the reagents needed to synthesize 1,3-butadiene starting from (a) 1,4-Dibromobutane (b) (c) (d) (e) (f) (g) HO Cl CI
-
Provide the reagents needed to accomplish transformations (a)-(g). More than one step may be necessary. OH OH OH OH OMe
-
Two companies, Energen and Hastings Corporation, began operations with identical balance sheets. A year later, both required additional manufacturing capacity at a cost of $50,000. Energen obtained a...
-
What is a centrally planned economy? Describe the link between central planning and communism.
-
A 0.2-lb model rocket is launched vertically from rest at time t = 0 with a constant thrust of 2 lb for one second and no thrust for t > 1 s. Neglecting air resistance and the decrease in mass of the...
-
Evans \& Sons, Inc. reported the following borrowings in a prior annual report: For each borrowing, indicate whether the bond or debenture was originally sold at its face value, a discount, or a...
-
Two eight-year-old boys were seriously injured when riding Honda mini trail bikes provided by their parents. The boys were riding on public streets and ran a stop sign when they were hit by a truck....
-
Hi TELFORD ENGINEERING P/L account: Menai $,000 P/L (before MEXIT) Menai $,000 Actual P/L (one year after MEXIT under outsource option) Sales 8,000 7,200 * Note: Exports to CETA based customers...
-
1. What mass of Cu(IO3) can be formed from 0.500 gram CuSO4 5H2O? 2. What mass of KIO3 is needed to convert the copper in 0.200 gram in CuSO4 5H2O to Cu(IO3)2? 3. What mass of AgI can be produced...
-
Predict the products of the following reactions. (a) (b) Eto OEt (1) EtMgBr (excess) (2) NH,CI, HO
-
Predict the products from each of the following reactions. (a) (b) (c) (d) (e) (f) (1) BH:THF (2) NaOH, HO
-
In Fig. 4.15 an astronaut is playing shuffleboard on Earth. The puck has a mass of 2.0 kg. Between the board and puck the coefficient of static friction is 0.35 and of kinetic friction is 0.25. (a)...
-
What role do race and ethnicity play in the social stratification of contemporary societies, and how can critical race theory help deconstruct the mechanisms that perpetuate racial and ethnic...
-
Wildhorse Company's accounting records reflect the following inventories: Dec. 31, 2022 Dec. 31, 2021 Raw materials inventory $240000 $190000 Work in process inventory 300000 160000 Finished goods...
-
Pool your time length data with data from other class members or the entire class so that the pooled data set has at least 100 observations. Designate someone in the group to calculate the mean and...
-
Braverman Company has two manufacturing departments-Finishing and Fabrication. The predetermined overhead rates in Finishing and Fabrication are $28.00 per direct labor-hour and 120% of direct...
-
Roger set his residence on fire for the insurance. While the fire was being fought, the police arrived to help with the crowds of onlookers. Roger realized he had done something wrong and went to one...
-
Henry Limited had investments in securities on its statement of financial position for the first time at the end of its fiscal year ended December 31, 2017. Henry reports under IFRS and its...
-
A local politician is concerned that a program for the homeless in her city is discriminating against blacks and other minorities. The following data were taken from a random sample of black and...
-
Name these compounds: CH3 a) CHCHCHCHCHCH, b) NHCH, NH
-
Draw structures for these compounds: (a) Diethyl ammonium bromide (b) N-Methyl-3-(1-methylpropyl)-2-octanamine
-
Name thesecompounds: CH3 CH3 T a) CHCH,CH_CHCHCH,CH, CHCH3 b) CHCH=CHCHCHCH3 CI c) CH,CH,CHC=CH d) e) f) g) OH CH3 H
-
The last four years of returns for a share are as follows: Year 1 2 3 4 Return 4.1% 27.8% 12.4% 4.3% a. What is the average annual return? b. What is the variance of the share's returns? c. What is...
-
This is a population standard deviation. The differences between sample standard deviation and population standard deviation are population standard deviation is calculated for a population, while...
-
Cloverdale Country Rodeo Inc. (CCRI) sells $1,000,000 of 10-year, 6% bonds for $980,500 plus accrued interest. The bonds are dated January 1, 2023, but due to market conditions are not issued until...

Study smarter with the SolutionInn App


