The 1H NMR signals for the aromatic hydrogens of methyl p-hydroxybenzoate appear as two doublets at approximately
Question:
The 1H NMR signals for the aromatic hydrogens of methyl p-hydroxybenzoate appear as two doublets at approximately 7.05 and 8.04 ppm (δ). Assign these two doublets to the respective hydrogens that produce each signal. Justify your assignments using arguments of relative electron density based on contributing resonance structures.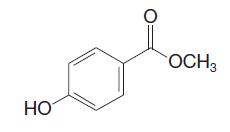
Transcribed Image Text:
HO 。 OCH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (14 reviews)
My argument on my assignment is as follows i The chemical shift of the downfield ...View the full answer

Answered By

Brian Otieno
I'm Brian , an experienced professional freelancer with countless hours of success in freelancing many subjects in different disciplines. Specifically, I have handled many subjects and excelled in many disciplines. I have worked on many Computer Science projects and have been able to achieve a lot in that field. Additionally, I have handled other disciplines like History, Humanities, Social Sciences, Political science, Health care and life science, and Religion / Theology. My experience generally in these subjects has made me able to deliver high-quality projects in a very timely fashion. I am very reliable at my job and will get the work done in time, no matter what. In Addition, I have managed to ensure that the work meets my client's expectations and does not cause an error. I am a hard-working and diligent person who is highly responsible for everything I do. Generally, Freelancing has made me more accountable for doing my job. Additionally, I have had a passion for writing for the last seven years in this field.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry
ISBN: 978-1118875766
12th Edition
Authors: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Question Posted:
Students also viewed these Sciences questions
-
Order the 1H NMR signals of the following compounds by chemical-shift position (lowest to highest). Which one is the most upheld? The most downfield? (a) H3C-CH3 (b) H2C==CH2 (c) H3C-O-CH3 (d) (e)...
-
The 1H NMR signal for bromoform (CHBr3) appears at 2065 Hz when recorded on a 300-MHz NMR spectrometer. (a) What is the chemical shift of this proton? (b) Is the proton in CHBr3 more shielded or less...
-
The 1H NMR signal for bromoform (CHBr3) appears at 2065 Hz when recorded on a 300-MHz NMR spectrometer. (a) What is the chemical shift of this proton? (b) Is the proton in CHBr3 more shielded or less...
-
Compare and contrast megaloblastic anemia caused by vitamin B12 deficiency and that caused by folic acid deficiency. (10)
-
Let M be the median (in points) and R be the range (in points) of four test scores (all in points), x1, x2, x3, and x4, where the scores are listed from smallest to largest. a. Write a formula for...
-
On a laboratory assignment, if the equipment is working, the density function of the observed outcome, X is Find the variance and standard deviation ofX. [2(1 -x), 0
-
Prepare a document flowchart to reflect how ANGIC Insurance Company processes its casualty claims. The process begins when the claims department receives a notice of loss from a claimant. Claims...
-
Lynch Co. uses the percentage of receivables basis to record bad debts expense and concludes that 2% of accounts receivable will become uncollectible. Accounts receivable are $400,000 at the end of...
-
-12 3. Scott deposits 80 today into an account that credits a nominal interest rate of 6% compounded quarterly. Roberta deposits 119.60 today into an account that credits a nominal discount rate of...
-
For this exercise, your client, Bright IDEAS Inc., has provided you with a listing of sales invoices. To test whether the client appears to have a receivables collectability problem, the auditor must...
-
The structure of thyroxine, a thyroid hormone that helps to regulate metabolic rate, was determined in part by comparison with a synthetic compound believed to have the same structure as natural...
-
The following reaction sequence was used by E. J. Corey (J. Am. Chem. Soc. 1969, 91, 56755677) at the beginning of a synthesis of prostaglandin F 2 and prostaglandin E 2 . Explain what is involved in...
-
The CNO is reviewing patient satisfaction benchmark data from their facility and four similar facilities (see table). What conclusions can they make, given the information? What are the limitations...
-
A USDCNH futures contract on HKEX covers USD100,000 with a minimum price change of $0.0001 is sold for CNH 6.6546. If you just place the initial margin of CNH11,305 and the maintenance margin is CNH...
-
Solve the triangle: If B = 100, C = 36.22, b = 5; Find c (round to the nearest whole number) Solve the triangle: If b = 3, a = 4.5, C = 20.74; Find c (round to the nearest whole number) Solve the...
-
When in which condition the promotional goal "to inform" is particularly important? explain.
-
When did Apple first enter the South Korea Market? What was Apple's marketing strategy to penetrate the Korean market? Provide citations.
-
Sarik was surprised by how many conversations it took to make a deal with CorpCo. What pitfall did Sarik experience? Select an answer: using improper anchors burning relationships low-ball...
-
McGilla Golf has decided to sell a new line of golf clubs. The clubs will sell for $750 per set and have a variable cost of $360 per set. The company has spent $150,000 for a marketing study that...
-
What is an insurable interest? Why is it important?
-
Explain whether these elimination reaction would be a good way to prepare thesealkenes: Cl H,O CH,OH + KOH CI EIOH PHCH=CHCH, b) PHCH CHCH; + NaOEt
-
Explain which of these reactions would provide a better synthesis of2-pentene: Br CH,OH CH,CH,CHCH,CH, + CH;0 CH,CH=CHCH,CH3 Br CH, CH CH=CHCH CH, CH,CHCH,CH,CH; + CH,0
-
Show the products of thesereactions: Br CH,OH CH3CH,CHCH,CH, + CH,0 CH,CH-CHCH,CH3 Br CH;OH CH,CHCH,CH,CH; + CH,0 CH.CH-CHCH,CH; CH, CH-CH2 2 b) ELOH Br Br
-
Request the audiovisual equipment well in advance. a. True b. False
-
Financial assets do not directly contribute to the capacity of the economy
-
On 1 May, you used internet banking to transfer $1,500 from your personal account into the bank account at Bank Sinatra that you just opened for your business. The funds were received instantly...

Study smarter with the SolutionInn App


