The following reaction sequence was used by E. J. Corey (J. Am. Chem. Soc. 1969, 91, 56755677)
Question:
The following reaction sequence was used by E. J. Corey (J. Am. Chem. Soc. 1969, 91, 5675–5677) at the beginning of a synthesis of prostaglandin F2α and prostaglandin E2. Explain what is involved in this reaction and why it is a reasonable process.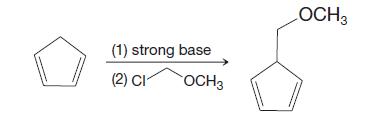
Transcribed Image Text:
(1) strong base (2) CI OCH 3 LOCH 3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (16 reviews)
Solution In the reaction sequence Grignard reagent MgBr is used to convert a secondary ...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry
ISBN: 978-1118875766
12th Edition
Authors: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Question Posted:
Students also viewed these Sciences questions
-
Explain what is involved in a put option.
-
In an attempt to make 1-chloro-l-cyclobutylpentane, the following reaction sequence was employed. The actual product isolated, however, was not the desired molecule but an isomer of it. Suggest a...
-
What is involved in estimating customer lifetime value (CLV)?
-
Problem 4.2 Ask the user to enter his/her age, with the prompt: "How old are you? Please enter your age as a number between 0 and 120. -> ". Check that what was entered is a number between 0 and 120....
-
Let x be the mean (in points) and s be the standard deviation (in points) of three test scores, x1, x2, and x3, all in points. a. Write a formula for the mean of the three test scores. b. Use the...
-
Referring to Exercise 4.35 find the mean and variance of the discrete random variable Z = 3X 2, when X represents the number of errors per 100 lines of code.
-
The Dewey Construction Company processes its payroll transactions to update both its payroll master file and its work-in-process master file in the same computer run. Both the payroll master file and...
-
Condensed financial data of Fulton Corp. for 2014 and 2013 are presented below. FULTON CORP. INCOME STATEMENT FOR THE YEAR ENDED DECEMBER 31, 2014 Sales ............... $15,200 Cost of goods sold...
-
Current Attempt in Progress Blossom Incorporated management is considering investing in two alternative production systems. The systems are mutually exclusive, and the cost of the new equipment and...
-
A feedback control system has the structure shown in Figure P2.35. Determine the closed-loop transfer function Y(s)/R(s) (a) By block diagram manipulation and (b) By using a signal-flow graph and...
-
The 1H NMR signals for the aromatic hydrogens of methyl p-hydroxybenzoate appear as two doublets at approximately 7.05 and 8.04 ppm (). Assign these two doublets to the respective hydrogens that...
-
Write mechanism arrows for the following step in the chemical synthesis by A. Robertson and R. Robinson (J. Chem. Soc. 1928, 14551472) of callistephin chloride, a red flower pigment from the...
-
Microwaves of wavelength 5.00 cm enter a long, narrow window in a building that is otherwise essentially opaque to the incoming waves. If the window is 36.0 cm wide, what is the distance from the...
-
When designing the packaging for your product, what represents all the elements you must consider?
-
What Money Can't Buy , Sandel elaborates on a number of examples where market logic and moral reasoning do not seem to align: tradeable procreation permits, purchasable pollution credits, the right...
-
All methods of judicial selection (the process by which judges gain their positions) face a tension between judicial independence and judicial accountability . On the one hand, it is generally...
-
David is a South African photographer who has worked for numerous media outlets over the past two decades as a freelancer. Recently, he was hired on a contract basis to cover the upcoming national...
-
After reading the Legacy of Court TV (Box 6.6) discuss the positive and negative effects of televising criminal trials. How does this compare to crime dramas that are focused on the courtroom....
-
You are considering a new product launch. The project will cost $875,000, have a four-year life, and have no salvage value ; depreciation is straight-line to zero. Sales are projected at 190 units...
-
Charles owns an office building and land that are used in his trade or business. The office building and land were acquired in 1978 for $800,000 and $100,000, respectively. During the current year,...
-
Show the products of thesereactions: CH, OH CH3 H,SO, H,SO, b) H3CCOH H PO, CH,CH-CHCH, H;SO4 a) CH
-
Show the products of thesereactions: CrO3 H,SO4 PCC a) b) . CH,Cl, acetone CrO3 2 pyridine PCC d) c) CH,Cl, CH,Cl, CH Ag,0 e) - THE
-
Show syntheses of these compounds from the indicated startingmaterials. a) CH,CH,CH,CH,N(CH3), from compounds with none of the CN bonds of the final product. CH3 b) CH,CH,OCHCH,CH; from alkyl halides...
-
The lessons from the crisis also suggest that monetary policy should lean against credit booms but not asset - price bubbles.
-
Question 4 XYZ was founded 10 years ago. It has been profitable for the last 5 years, but it has needed all of its earnings to support growth and thus has never paid a dividend. Management has...
-
Summarize the pros and cons of judgmental and empirically based credit evaluation systems. What would you recommend be the primary evaluation system for the loans in the following table? Complete the...

Study smarter with the SolutionInn App


