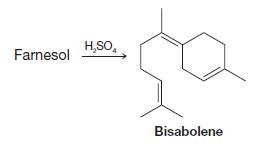
When farnesol is treated with sulfuric acid, it is converted to bisabolene. Outline a possible mechanism for
Question:
When farnesol is treated with sulfuric acid, it is converted to bisabolene. Outline a possible mechanism for this reaction.
Transcribed Image Text:
Farnesol H₂SO4 Bisabolene
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 77% (9 reviews)
Solution The proposed mechanism for farnesol to bisabolene ...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry
ISBN: 978-1118875766
12th Edition
Authors: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Question Posted:
Students also viewed these Sciences questions
-
When 2-methyl-2-propanol is treated with sulfuric acid, 2-methylpropene is formed. Show all of the steps in the mechanism for this reaction. Don't forget to use curved arrows to show the movement of...
-
A possible mechanism for the pathology of trinucleotide repeat diseases is aberrant translation in which all three reading frames are used. Which repeating amino acid residues will result from the...
-
A possible mechanism for a gas-phase reaction is given below. What is the rate law predicted by this mechanism? (fast equilibrium) 2NOCI NOC NO2NOCI (slow)
-
Suppose that you are holding your toy submarine under the water. You release it and it begins to ascend. The graph models the depth of the submarine as a function of time. What is the domain and...
-
Repeat Exercise 29, but let n be the number of collisions (in thousands) for the year that is t years since 1985. Which of your responses for this exercise are the same as those for Exercise 29?...
-
For a single degree of freedom spring-mass-damper system subject to forced harmonic vibration, the following FRF was measured (two figures are provided with different frequency ranges). Using the...
-
When would you use a motion to strike?
-
1. What are the internationalisation drivers Walmart International has struggled with? 2. What might be the dangers for a large Western retailer in staying out of emerging markets?
-
The Three Key Themes in Strategic Cost Management are: i. Value Chain Analysis, ii. Strategic Positioning Analysis, and iii. Cost Driver Analysis REQUIRED: Discuss how these are applied in the...
-
Rose-comb chickens mated with walnut-comb chickens produced 15 walnut-, 14 rose-, 5 pea-, and 6 single-comb chicks. Determine the genotypes of the parents.
-
When morphine reacts with 2 mol of acetic anhydride, it is transformed into the highly addictive narcotic heroin. What is the structure of heroin?
-
One of the important steps in Gates synthesis of morphine involved the following transformation: Suggest how this step was accomplished. CHO CHO NC CHO CHO NC-
-
Refer to the data on annual mortgage interest rate (Yt), Exercise 14.41. You fit the simple linear regression model, E(Yt) = 0 + 1t, to the data for the years 1990-2015 (t = 0, 1, 2, c, 25). a. Find...
-
80. Among the following most stable carbocation is (1) CH (2) CH CH3 + (3) (4) CH2=CH-CH2 CH3
-
On March 3, 2020, the Board of Governors of the Federal Reserve System unexpectedly decided to cut interest rates (the Federal Funds Rate) by 50 basis points to a target range of 1-1.25%. At the same...
-
3) Ben and Jerry are movers for Super Duper Moving company. They are arguing about the need for the ramp. Ben says that they do not need the ramp because the height to the truck is the same, so they...
-
You have just been hired by FAB Corporation, the manufacturer of a revolutionary new garage door opening device. The president asked you to review the company's costing system and "do what you can to...
-
72. For a chemical reaction if rate constant is 2 103 mol L min then order of reaction is :- (1) Zero (2) 1 (3) 2 (4) Can not predict 73. Which of the following is not an electrophile? (1) CH3 (3) C...
-
The major stock market indexes had strong results in 2012. The mean one-year return for stocks in the S&P 500, a group of 500 very large companies, was +13.41%. The mean one-year return for the...
-
Use multiplication or division of power series to find the first three nonzero terms in the Maclaurin series for each function. y = e x2 cos x
-
Predict the multiplicity of the absorption for Hm if Jam = Jmx. Explain. , , . -C-C-C-
-
Construct a tree diagram for the absorption of Hm assume that Jam . . - -C-
-
Predict the multiplicities of the absorptions for the hydrogen's of these groups, assume that hydrogen's labeled a are different from those labeled x but that all of those labeled a are identical and...
-
Matrix B is given as follows, -18 20 -61 B-12 14 -3 30 -30 13. a) Use the Gerschgorin's Circle Theorem to determine a region containing all the eigenvalues of B. b) Find the dominant eigenvalue (21)...
-
5.63. Acetone can be removed from acetone-air mixtures using simple counter- current cascades, by adsorption onto charcoal (Foust et al. 1980). We wish to find the required number of equilibrium...
-
Use characteristic polynomial to find the eigen value and eigen vector for A S

Study smarter with the SolutionInn App


