A compound of formula C 11 H 16 N 2 gives the IR 1 H NMR, and
Question:
A compound of formula C11H16N2 gives the IR 1H NMR, and 13C NMR spectra shown. The proton NMR peak at δ2.0 disappears on shaking with D2O Propose a structure for this compound, and show how your structure accounts for the observed absorptions.
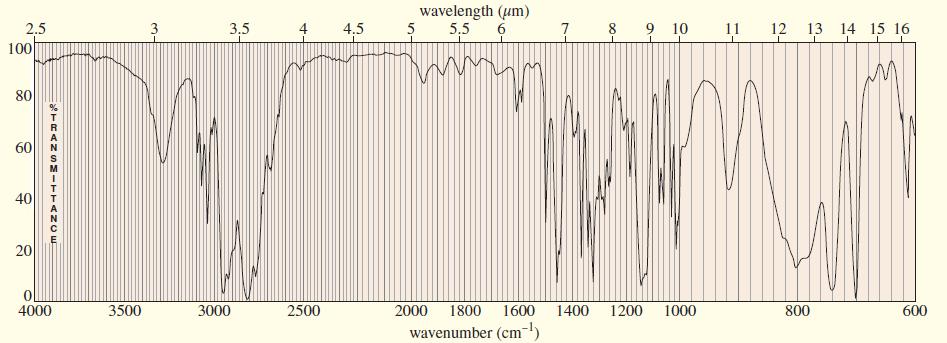
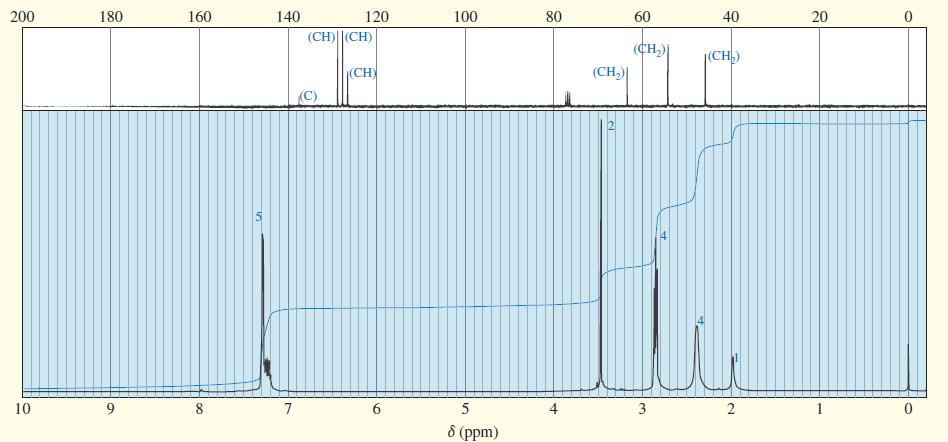
Transcribed Image Text:
wavelength (um) 2.5 3.5 4.5 5 5,5 6 7 8 9 10 11 12 13 14 15 16 100 80 60 40 4000 3500 3000 2500 2000 1800 1600 1400 1200 1000 800 600 wavenumber (cm-) is 4. TRANS=-TTAN OE 20
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 81% (11 reviews)
Molecular formula C 11 H 16 N 2 Double bond equivalent DBE of the given compound is 11162 5 Therefor...View the full answer

Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
An unknown compound of molecular formula C5H9NO gives the IR and NMR spectra shown here. The broad NMR peak at δ7.55 disappears when the sample is shaken with D2O. Propose a structure,...
-
An unknown compound shows a weak molecular ion at m/z87 in the mass spectrum, and the only large peak is at m/z30. The IR spectrum follows. The NMR spectrum shows only three singlets: one of area 9...
-
An unknown compound gives the NMR, IR, and mass spectra shown next. Propose a structure, and show how it is consistent with the observed absorptions. Show fragmentations that account for the...
-
To meet the demand for parking, your town has allocated the area shown here. As the town engineer, you have been asked by the town council to find out if the lot can be built for $11,000. The cost to...
-
The cumulative formation constant for SnCl2(aq) in 1.0 M NaNO3 is 2 = 12. Find the concentration of SnCl2(aq) for a solution in which the concentrations of Sn2+ and Cl- are both somehow fixed at 0.20...
-
During your audit of Patti Companys ending inventory at December 31, 20X1, you find the following inventory accounting errors: a. Goods in Pattis warehouse on consignment from Valley, Inc., were...
-
This chapter placed emphasis on a "transaction-based perspective" related to customers. What would be different about a transaction involving an investor? What do we mean by transaction?
-
Shiloh Acres sponsors a defined-benefit pension plan. The corporations actuary provides the following information about the plan. The average remaining service life per employee is 10 years....
-
What are the total industry costs of pollution control (i.e., for both firms combined) if a uniform emission standard is utilized to achieve an aggregate emission reduction of 6 tons? In other words,...
-
The above reaction involves two sequential Heck reactions and three organopalladium intermediates. In the box below draw the structure of intermediate C. ? You do not have to consider...
-
Propose mechanisms for the following reactions. a. b. c. d. e. f. CH,OH, H* CH;O OCH3 Ph-C-H Ph
-
Show how you might synthesize the following tertiary amine three different ways, each using a different secondary amine and adding the final substituent by (a) Reductive amination (3 ways). (b)...
-
Record each transaction of Roses Flower Shop. a. Purchased merchandise for sale on October 1 for $3,600, to be paid by October 30. b. Sold merchandise for $900 cash on October 3. The merchandise cost...
-
Make a content inventory for an organizational website with which you are familiar. List the content types present on the site and, as best you can, who in the organization (by role, if not by name)...
-
Hannah is purchasing chocolate truffles for her mother and nana on Mother's Day. Chocolate truffles come in boxes of 6 pieces each and milk chocolate truffles come in boxes of a dozen. If she can...
-
Consider a technical communication project you've completed, or one that remains due. Which of Schriver's audience model(s) (classification-driven, intuition-driven, feedback-driven) most closely...
-
How does our accepting that contemporary knowledge evolves out of our interaction with and between technology and the real world help us understand the role of information overload in our...
-
What information could one gather to illustrate the influence of tools on technical communication practice?
-
How will the total cost of borrowing be affected if a bond is sold (a) At a discount and (b) At a premium?
-
What is the back work ratio? What are typical back work ratio values for gas-turbine engines?
-
Give a curved-arrow mechanism for the biosynthesis of the carbocation intermediate from geranyl pyrophosphate.
-
How Geranyl pyrophosphate is converted into farnesyl pyrophosphate. OH farnesol
-
Give the principal product(s) expected when 4-methyl-cyclohexene or other compound indicated reacts under the conditions in Problem 17.18. (a) Br2 in CH2Cl2, dark (b) A-bromosuccinimide in CCl4,...
-
Image transcription text You encounter a split system that uses R-22 refrigerant and observe the following refrigeration parameters from the unit's control display. The unit is operating in cooling...
-
Calculating Present Values Imprudential, Inc., has an unfunded pension liability of $645 million that must be paid in 25 years. To assess the value of the firm's stock, financial analysts want to...
-
Describe the operating activities of each company noting similarities and difference between COCA COLA & PEPSICO. Identify two economy wide factors and industry wide factors that could impact on the...

Study smarter with the SolutionInn App


