Ketones react with dimethyl sulfonium methylide to yield epoxides by a mechanism that involves (1) an initial
Question:
Ketones react with dimethyl sulfonium methylide to yield epoxides by a mechanism that involves (1) an initial nucleophilic addition followed by (2) an intramolecular SN2 substitution. Show the mechanism.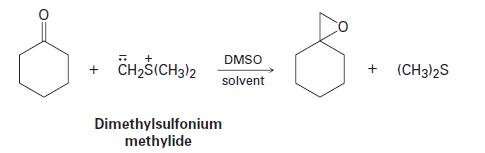
Transcribed Image Text:
+ CH2S(CH3)2 DMSO solvent Dimethylsulfonium methylide + (CH3)2S
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
lets go through the mechanism for the reaction of a ketone with dimethyl sulfonium methylide to yiel...View the full answer

Answered By

User l_1006857
I am a computer science professional with expertise in databases, AI programming, data structures and algorithms, and mathematics. With a strong background in these areas, I possess the knowledge and skills necessary to design and optimize database systems, develop intelligent algorithms and models, and solve complex computational problems. My proficiency in SQL, NoSQL, machine learning techniques, and mathematical concepts equips me to contribute to innovative projects and drive technological advancements.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Proteins can be cleaved specifically at the amide bond on the carboxyl side of methionine residues by reaction with cyanogen bromide, BrC = N. The reaction occurs in several steps: (a) The first step...
-
Ketones and aldehydes react with primary amines to give imines. They react with secondary amines to give enamines (vinyl amines). (a) For review, propose a mechanism for the following formation of an...
-
Show how you might use a nucleophilic substitution reaction of 1-bromopropane to synthesize each of the following compounds. (You may use any other compounds that are necessary.) (a) (b)...
-
If a zero of f is i, find the complete factored form of f(x) = x 4 + x 3 + 2x 2 + x + 1.
-
Identify a restaurant where you have dined. Identify some of the types of costs that the restaurant incurs in each of the four cost-of-quality categories. Consider how those costs are related to each...
-
Calculate the bandwidth of the light for the following wavelength ranges (assume a propagation speed of 2 10 8 m): a. 1000 to 1200 nm b. 1000 to 1400 nm
-
Robert Shapiro was the owner and CEO of Woodbridge, a supposed investment firm. Woodbridges main business model was to solicit money from individuals to invest in low-risk and conservative...
-
(EPS: Simple Capital Structure) On January 1, 2010, Chang Corp. had 480,000 shares of common stock outstanding. During 2010, it had the following transactions that affected the common stock account....
-
Find all the complex numbers z such that z += 0. Is the set open or closed? {zC: z*+i=0}
-
Reaction of butan-2-one with HCN yields a cyanohydrin product [R 2 C(OH)CN] having a new chirality center. Explain why the product is not optically active.
-
Give IUPAC names for the following acyl derivatives: (a) (d) CH3 CH3CHCH2CH2CCI (g) 0 0=0 H2C = CHCH2CH2CNHCH3 (b) (e) (h) CHCNH2 || OCHCH3 CH3 CH3 CH3CH2CHCN (c) (f) CH3CHCOCHCH3 CH3 CH3 CH3 CHCH3
-
Distinction between avoidable costs and cost behavior Preston Phones, Inc., makes telephones that it sells to department stores throughout the United States. Preston is trying to decide which of two...
-
What is a dependency relationship? How do you identify them?
-
Assuming time and money were not important concerns, would BPR projects benefit from additional time spent understanding the as-is system? Why or why not?
-
What are the main building blocks for the sequence diagram? How are they represented on the model?
-
What is meant by balancing the models?
-
Contrast the following sets of terms: state, behavior, class, object, action, and activity.
-
The Ashland Company recently hired you to review its control procedures for the purchase, receipt, storage, and issuance of raw materials. You prepared the following comments, which describe Ashlands...
-
Before the latest financial crisis and recession, when was the largest recession of the past 50 years, and what was the cumulative loss in output over the course of the slowdown?
-
Consider the pK a values of the following constitutional isomers: Using the rules that we developed in this chapter (ARIO), we might have expected these two compounds to have the same pKa....
-
Consider the following compound with molecular formula C 4 H 8 O: (a) Draw a constitutional isomer that you expect will be approximately one trillion (10 12 ) times more acidic than the compound...
-
There are only four constitutional isomers with molecular formula C 4 H 9 NO 2 that contain a nitro group (-NO 2 ). Three of these isomers have similar pK a values, while the fourth isomer has a much...
-
Y8 You invested $85,000 in a mutual fund at the beginning of the year when the NAV was $33.97 . At the end of the year, the fund paic $0.39 in short-term distributions and $0.56 in long-term...
-
3. (20%) The dc machine (12kW, 100V, 1000 rpm) is connected to a 100V dc supply and is operated as a dc shunt motor. At no-load condition, the motor runs at 1000 rpm and the armature takes 6 amperes....
-
b) As the engineer for your company, you have been assigned to design a pipeline system. The information given to you is that the pipeline is horizontal and has a pipe with a diameter of 0.6 meters....

Study smarter with the SolutionInn App


