There are three resonance structures of naphthalene, of which only one is shown. Draw the other two.
Question:
There are three resonance structures of naphthalene, of which only one is shown. Draw the other two.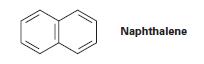
Transcribed Image Text:
Naphthalene
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (7 reviews)
ic aromatic hydrocarbon consisting of two benzene rings fused toge...View the full answer

Answered By

Surendar Kumaradevan
I have worked with both teachers and students to offer specialized help with everything from grammar and vocabulary to challenging problem-solving in a range of academic disciplines. For each student's specific needs, I can offer explanations, examples, and practice tasks that will help them better understand complex ideas and develop their skills.
I employ a range of techniques and resources in my engaged, interesting tutoring sessions to keep students motivated and on task. I have the tools necessary to offer students the support and direction they require in order to achieve, whether they need assistance with their homework, test preparation, or simply want to hone their skills in a particular subject area.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Look at the three resonance structures of naphthalene shown in Section 15.7, and account for the fact that not all carboncarbon bonds have the same length. The C1C2 bond is 136 pm long, whereas the...
-
The organic molecules shown here are derivatives of benzene in which six-member rings are "fused" at the edges of the hexagons. (a) Determine the empirical formula of benzene and of these three...
-
Draw three resonance structures of sulfur dioxide (SO2). Indicate the most plausible structure(s).
-
. 4. Bank overdrafts repayable on * 1 point demand may be included in the cash and cash equivalent balance. True O False
-
The account balances for Pauls Furniture, Inc., for the year ended August 31, 2012, are presented next in random order: Requirements 1. Prepare Pauls Furnitures single-step income statement. 2. Would...
-
1. Assess how Walmart is managing ethics and social responsibility as one of the largest corporations in the world. 2. Evaluate various ethical issues Walmart has faced and how the company responded...
-
Discuss the themes, theory, and/or phenomenon that would be anticipated to emerge as a result of the examination. Develop a hypothetical research scenario that would necessitate the use of the Action...
-
The trial balance for the Venice Beach Kite Shop, a business owned by Molly Young is shown on page 550. Year-end adjustment information is as follows: (a and b) Merchandise inventory costing $35,000...
-
Why is it important to learn SQL? In which jobs you need to know SQL? 2A- What is the definition of DML and DDL? Give examples in each case. 2B- Consider the following two tables below: VENDOR and...
-
Show the structure of Teflon by drawing several repeating units. The monomer unit is tetrafluoroethylene, F 2 C=CF 2 .
-
Draw and name the products you would expect to obtain by reaction of the following substances with Cl 2 and FeCl 3 (blue=N, reddish brown=Br): (a) (b)
-
Consider a finite-duration sequence (a) Sketch the sequence s (n) with six-point DFT S(k) = W 2 * X (k) k = 0, 1 6 (b) Determine the sequence y (n) with six-point DFT Y (k) = Re |X (k)|. (c)...
-
Byron Bright, an inventor, sells the patent rights on his latest invention to Wilson Corporation. Wilson intends to manufacture and sell Byrons invention. Byron will receive $50 per unit Wilson sells...
-
A consumer rights organization wanted to check out different diet plans. It recruited 33 volunteers and sent them to four different programs. After the first 2 weeks, the weight losses, in pounds,...
-
Explain the difference between a population and a subpopulation.
-
Kirk Kelley, a single taxpayer, was engaged in the following transactions in 2019: Kirk lent a friend $2,500; that person did not pay the loan when it was due and then declared bankruptcy. The loan...
-
Cathy Crafts grants Dan Deputy a franchise to sell cards and gifts. Dan pays Cathy a fee of $1,000 per month plus a percentage of the monthly profits. Dan must purchase his supplies from Cathy and...
-
Teeds Manufacturing Corporation has the following shareholders equity at December 1, 2016: Shareholders Equity Share capital $2 preferred shares, no par value, cumulative....... 20,000 Shares...
-
In Exercises 1558, find each product. (9 - 5x) 2
-
Which of the following compounds show cis-trans isomerism? Draw the cis and trans isomers of those that do. a. CHF=CHF b. F 2 C=CH 2 c. CH 2 =CH-CH 2 -CH 3 d. e. f. CHCH,
-
Give the relationship between the following pairs of structures. The possible relationships are: same compound cis-trans isomers constitutional isomers (structural isomers) not isomers (different...
-
The C=O double bond has a dipole moment of about 2.4 D and a bond length of about 1.23 . (a) Calculate the amount of charge separation in this bond. (b) Use this information to evaluate the relative...
-
Two mutually exclusive projects are under consideration with the details shown. The company's required rate of return for projects of this risk level is 15%. using this information, answer the...
-
Summarize an article: Title: New Conceptual Foundations for Islamic Business Ethics: The Contributions of Abu-Hamid Al-Ghazali By: Yusuf Sidani Akram Al Ariss (2014) J Bus Ethics (2015) 129:847-857
-
Peace Inc. is a very small-scale producer of holiday season paper napkins located in central Texas, operating primarily around the Holiday season. Peace's entire annual production takes place over a...

Study smarter with the SolutionInn App


