(a) Organolithium reagents such as methyllithium (CH 3 Li) react with carboxylic acids to give ketones. Two...
Question:
(a) Organolithium reagents such as methyllithium (CH3Li) react with carboxylic acids to give ketones. Two equivalents of the lithium reagent are required, and the ketone does not react further. Suggest a mechanism for this reaction that accounts for these facts.
(b) Give the product of the reaction given in part (b) of Fig. P20.51.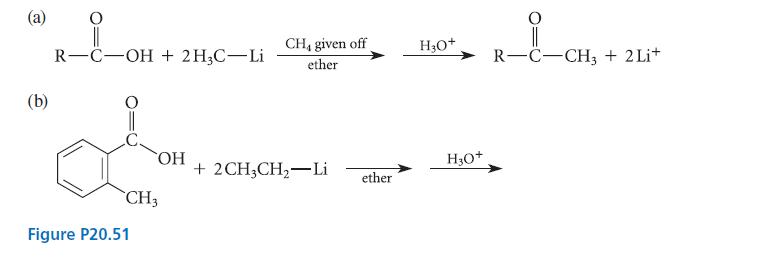
Transcribed Image Text:
(a) (b) R-C-OH + 2H3C-Li OH CH3 Figure P20.51 CH, given off ether + 2CH3CH₂ Li ether H3O+ H3O+ R-C-CH3 + 2Li+
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (6 reviews)
a The hint refers to the fact that organolithium reagents like Grignard reagents are stron...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The racemization of amino acids is an important reaction in a number of bacteria. This is a pyridoxal-phosphate catalyzed reaction. Outline a curved-arrow mechanism for this reaction showing clearly...
-
The reaction of nitric oxide (NO(g)) with molecular hydrogen (H2(g)) results in the production of molecular nitrogen and water as follows: 2NO(g) + 2H 2 (g) N 2 O(g) + 2H 2 O(g) The experimentally...
-
Tert-butyl esters can be prepared by the acid-catalyzed reaction of methylpropene (isobutylene) with carboxylic acids. Suggest a mechanism for this reaction that accounts for the role of the acid...
-
Since the introduction of enhanced security measures by the U.S. Department of Homeland Security in 2017, flights bound for the U.S. from Canada are subject to additional screening by airline...
-
Review the data in Short Exercise 5-10. Requirement 1. Prepare Carolina Communications classified balance sheet at July 31, 2012. Use the report format.
-
A bank has the following balance sheet: Suppose interest rates rise such that the average yield on rate- sensitive assets increases by 45 basis points and the average yield on rate- sensitive...
-
In the benzene adsorber of Example 9.7, the flow rate is increased to \(0.25 \mathrm{~m}^{3} / \mathrm{s}\). Calculate the breakthrough time and the fraction of the bed adsorption capacity that has...
-
The chart of accounts of Lopez Company includes the following selected accounts. 112 Accounts Receivable 401 Sales 120 Merchandise Inventory 412 Sales Returns and Allowances 126 Supplies 505 Cost of...
-
Pawan is pursuing MBA and shares a room with you in the private hostel. He desperately needs a job after completing his course because of educational loan and weak economic background. In his last...
-
Give a curved-arrow mechanism for each of the reactions given in Fig. P20.52. (a) (b) OEt 1 H3C-C-OEt + HO T OEt an orthoester (c) HC H3C C-CH3 COH dil. HCl (catalyst) CH3OH + C=CH + :C=O: dil. HCI...
-
In each case, draw the structure of the cyclic anhydride that forms when the dicarboxylic acid is heated. (a) (b) (c) COH 4 COH COH meso-a,-dimethylsuccinic acid COH
-
A company starts in business on 1 January 2014, the financial year end being 31 December. You are to show: (a) The equipment account. (b) The provision for depreciation account. (c) The statement of...
-
a) Using integration by parts, show that 1(x)= Hint: 1xt and te has an easy antiderivative. b) Repeat this logic again to show that 1 21-dr. 1(x)=-2 + c) Repeat twice more to start to notice a...
-
E-commerce store dealing in traditional clothing in Canada, suggest some plans and techniques for Future Growth & Expansion: 1. Plan how to grow the company and the product line (give real examples...
-
A proposed new venture will cost $85,000 and should produce annual cash flows of $30,000, $55,000, $40,000, and $40,000 for Years 1 to 4, respectively. The discount rate is 10 percent. What is the...
-
What are ways in which a monopolist can engage in price discrimination? Provide one or two examples of price discrimination you observe in your daily life? What type of price discrimination is it?
-
Suggest how you might respond to the Ma's concern that Zian is rejecting his Chinese culture. This morning you notice that Zian starts a conversation with one of his peers, how could you share this...
-
What information is conveyed by a cost-volume profit graph in addition to a companys break-even point?
-
Listed below are several terms and phrases associated with basic assumptions, broad accounting principles, and constraints. Pair each item from List A (by letter) with the item from List B that is...
-
Provide the reagents necessary to achieve the following synthetic transformations. More than one step may be required. (a) (b) (c) (d) (e) CN CN HOOG Br
-
Explain the product distribution below based on the polarity of the diene and dienophile, as predicted by contributing resonance structures for each. OCHso OCH heat OCH+ Major (plus enantiomer) Minor...
-
Mixing furan (Problem 13.45) with maleic anhydride in diethyl ether yields a crystalline solid with a melting point of 125(C. When melting of this compound takes place, however, one can notice that...
-
9. Determine the next three terms of the sequence. Explain your reasoning. (4 marks) a. 1 2 3 4 2'5'8'11 b. 1, -8, 27, -64, 125......... 10. A population of rabbits if left unchecked will triple...
-
Question 2 A new method of packaging for a drug, making use of hermetically sealed capsules containing an oxygen-free gas mixture is being developed, to try to extend the shelf life of a preparation...
-
The volume in a water tank can be modeled by the function V (t) = -2t^2 + 5t + 7, where V is the volume in gallons and t is the time after 12: 00 pm on a particular day. Explain how you would...

Study smarter with the SolutionInn App


