Give an IUPAC name for each of the following compounds, which may have been isolated from the
Question:
Give an IUPAC name for each of the following compounds, which may have been isolated from the shoes of a tennis player. Ignore stereochemistry in (a).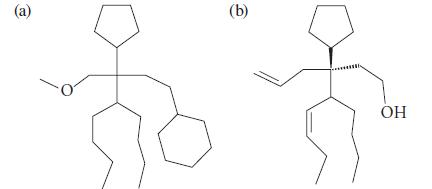
Transcribed Image Text:
(a) (b) HO
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
The IUPAC names for the compounds in the image are Compound a 2hydroxy2methylpropanoic acid also kno...View the full answer

Answered By

Kennedy Odhiambo
As a professional writer, I have been in the field for over 5 years having worked as a lecture in different tertiary institutions across the world. With this impeccable experience, I assure provision of a good and supporting environment for students to learn.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Give the IUPAC name for each of the following compounds: (a) CH3(CH2)25CH3 (b) (CH3)2CHCH2(CH2)14CH3 (c) (CH3CH2)3CCH(CH2CH3)2 (d) (e) (f) (g)
-
Give the IUPAC name for each of the following alkyl groups, and classify each one as primary, secondary, or tertiary: (a) CH3(CH2)10CH2-- (b) (c) --C(CH2CH3)3 (d) (e) (f) -CH2CH2CHCH2CH2CH3 CH2CH3...
-
Give the IUPAC name for each of the following compounds. a. b. CH C-CHCHCH CH CH2 CH3 CH CH2CH2CHCH2CH2CH3 CH CH2
-
You are the CEO of Green Paper Inc., a producer of high-end printing paper with an emphasis on environmentally friendly "green" production methods. One of your employees has proposed a significant...
-
Why is a confirmed LOC even more attractive to a seller than one that is not confirmed?
-
The Internet is a useful resource for gathering tax information. An important aspect of taxes discussed in this chapter is how deductible IRAs and Roth IRAs can be used to save for retirement. One...
-
White Company can invest in one of two projects, TD1 or TD2. Each project requires an initial investment of $101,250 and produces the year-end cash inflows shown in the following table. Required 1....
-
Suppose Nordstrom, Inc., which operates department stores in numerous states, has the following selected financial statement data for the year ending January 30, 2014. For the year, net sales were...
-
Various tools are available for developing and managing an auditing project plan and associated elements of an audit. Which tool is designed to enable an auditor to track audit deficiencies and areas...
-
For the following problems, see Table 8.2 for structures and dipole moments. Explain your reasoning in each case. (a) With which one of the following solvents is DMSO not miscible: water, acetone,...
-
Thiols of low molecular mass are known for their extremely foul odors. In fact, the following two thiols are the active components in the scent of the skunk. Give the IUPAC substitutive names for...
-
Shen Co. reports the costs incurred below for the month ended May 31. The company has no beginning Work in Process Inventory. Overhead is applied using a predetermined overhead rate of 120% of direct...
-
What is the benchmark for each ratio? Why is it important to use benchmarks or historical numbers for comparison? Are Marabelle's ratios falling within appropriate range? Explain. What elements does...
-
If the equilibrium price of concert tickets were $40, a maximum price of $40 will have no effect on the number of people who attend classical music concerts. Explain
-
Two college students, John and James, are drinking beer at a local bar. John asks James to help him paint the public statute pink to protest the lack of progress in cancer research. Such conduct is a...
-
The country of Bolivia had a Gross Domestic Product of $79 billion in 2016 and a population of 11 million people,What would be the GDP per capita ?
-
The price at which sellers are not willing to offer a good is $80 per unit. As the quantity supplied of the good increases by one unit, the minimum price at which someone is willing to sell that unit...
-
Assume a firm is a monopsonist that can hire its first worker for $6 but must increase the wage rate by $3 to attract each successive worker. Draw the firms labor supply and marginal resource cost...
-
Which of the following statements is false? a. Capital leases are not commonly reported in a Capital Projects Fund. b. A governmental entity may report a Capital Project Fund in one year but not the...
-
Some NMR spectrometers operate at 4 x 108Hz (400MHz), what is the energy of this radiation?
-
Explain which of these bonds has the absorption for its stretching vibration at higher wave number: (a) C H or C D (b) C = C or C C (c) C C1 or C I
-
Indicate the positions of the absorption bands and any other noteworthy features in the hydrogen region of the IR spectra of thesecompounds: CH3 ) C,CH b) NH2 d) CH2=CHCH,CH,OH f) CH,CH,CH,CH e)...
-
Hello, Could you help me to resolve this exercice please ? Thanks a lot a) Company A is expected to distribute a dividend of (3 per share in two months. Calculate the price of a six-month European...
-
Employment and labour law course
-
1) What are the common characteristics among Canadian labour relations laws? Specifically, what do the Canada Labour Code Part I and III have in common with the Labour Relations Act in Manitoba and...

Study smarter with the SolutionInn App


