Thiols of low molecular mass are known for their extremely foul odors. In fact, the following two
Question:
Thiols of low molecular mass are known for their extremely foul odors. In fact, the following two thiols are the active components in the scent of the skunk. Give the IUPAC substitutive names for these compounds.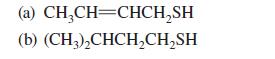
Transcribed Image Text:
(a) CH₂CH=CHCH₂SH (b) (CH3)₂CHCH₂CH₂SH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
ANSWER a The IUPAC substitut...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The end-of-year parties at Yearling, Inc., are known for their extravagance. Management provides the best food and entertainment to thank the employees for their hard work. During the planning for...
-
The end-of-year parties at Golden Beach, Inc., are known for their extravagance. Management provides the best food and entertainment to thank the employees for their hard work. During the planning...
-
Case Study: The Container Store: An Employee-Centric Retailer Introduction : It seems odd that a store would choose to specialize in selling containers and storage units, let alone make a profit from...
-
3. How do the marginal costs of pollution reduction and the marginal costs of pollution damage change as pollution levels increase? The marginal costs of pollution reduction remain constant and the...
-
In the Centrifugal Casting case, what international event made the LOC invaluable to the seller?
-
On January 1, 20X1, Alpha Corporation acquired all of Bravo Companys assets and liabilities by issuing shares of its $3 par value stock to the owners of Bravo Company in a business combination. Alpha...
-
For each of the following residual plots, determine whether the assumptions of the linear model are satisfied. If they are not, specify which assumptions are violated. b. C. d.
-
1. Name the constraints and indicate whether each is present, future, internal, external, mandatory, or desirable. 2. Explain why it is important to define the payroll projects scope. Explain how to...
-
You want to deposit $10,000 in a bank certificate of deposit (CD). You consider the following banks: Bank A offers 5.85% annual interest compounded annually. Bank B offers 5.75% annual interest rate...
-
Give an IUPAC name for each of the following compounds, which may have been isolated from the shoes of a tennis player. Ignore stereochemistry in (a). (a) (b) HO
-
Give the IUPAC substitutive name for each of the following compounds, which have been used as general anesthetics. (a) Br I H-C-CF3 I Cl halothane (b) ClCH-CF-OCH3 methoxyflurane
-
What consequence does the lack of uniqueness of polar coordinates have for graphing? Give an example.
-
1 2 3 For the matrix A = 0 45 find the determinant, cofactor matrix, and inverse. 2 -1 1 (a) [2 points] Find det A. (b) [4 points] Find the cofactor matrix C. (c) [4 points] Use the cofactor matrix...
-
One-hundred milliliters of 1.75 mM NaCl is mixed with 80.0mL of3.20 mM MgCl2 at 20 degrees celcius. Calculate the osmotic pressureof each starting solution and that of the mixture, assuming thatthe...
-
A firm's investments cost $100,000 and are expected to return $118,000 before taxes at the end of 1 year at which point it will be closed down. The firm pays taxes at the marginal rate of 40%, and...
-
A 10-year, 7%, $100,000 bond is sold by Richardson Incorporated. The bond pays interest semi-annually. The market rate of interest for comparable bonds is 6%. At what price does the bond sell? Round...
-
3. Consider a linear regression model: yi x' = (Xi1, ..., Xik) and B = (B,..., k)'. (i) Show that E(xu) = 0 (show how you obtain the result, explain your proof). (ii) Replacing ui by ui = yi - x' in...
-
An Internal Revenue Oversight Board survey found that 82% of taxpayers said that it was very important for the Internal Revenue Service (IRS) to ensure that high-income tax payers do not cheat on...
-
Assume today is the 21st of February. Using the information below, FT Extract, answer the following questions (parts i and ii). You work for a US company that is due to receive 250 million in June...
-
Explain why the presence of a triple bond is much easier to detect in the IR spectrum of 1-hexyne than it is in the spectrum of3-hexyne. 80 60 O The sp-hybridized CH absorption bands: from 3000-2850...
-
The exhaust from a poorly maintained automobile may contain a wide variety of different hydrocarbon pollutants. Why is the 3000 to 2900 cm1 region a good place to monitor the amount of these...
-
Predict the positions of the absorption bands in the IR spectra for the carbonyl groups of thesecompounds. HO. b) CH,CH-CHCH (p
-
Burch Company Income Statement For the year ended December 31 Sales $250,000 Cost of goods sold (160,000) Depreciation expense (26,400) Other expenses (35,000) Income tax expense (12,000) Net income...
-
AMB Bhd is an international industrial products manufacturer in Malaysia. The company's financial statements ended 31 December 2021 were authorized for issue by board of directors on 30 April 2022....
-
Use the following information to calculate the net cash provided or used by financing activities. (a) Paid $28,200 cash to settle long-term notes payable at its $28,200 maturity value. (b) Acquired...

Study smarter with the SolutionInn App


