Give the structures of the principal organic product(s) expected in each of the reactions given in Fig.
Question:
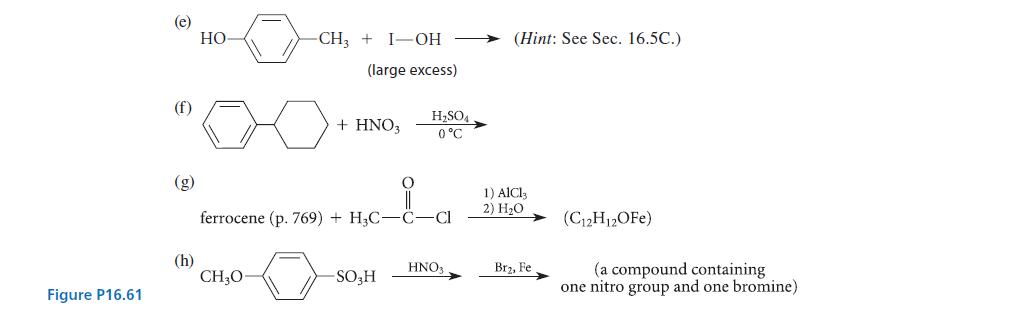
Give the structures of the principal organic product(s) expected in each of the reactions given in Fig. P16.61, and explain your reasoning.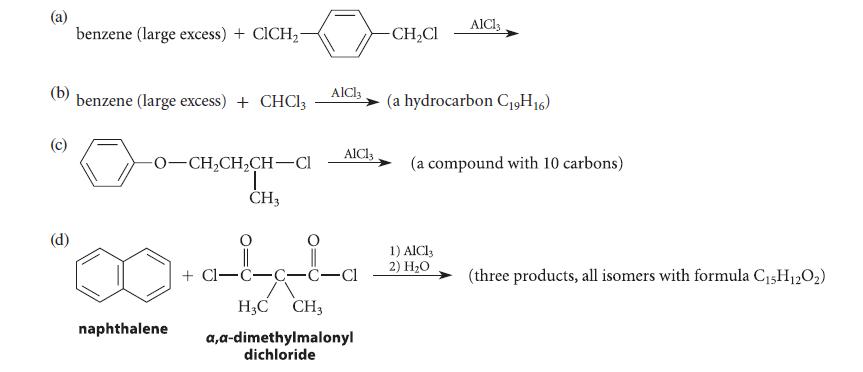
Transcribed Image Text:
(b) (d) benzene (large excess) + ClCH₂- benzene (large excess) + CHCl3 -0-CH₂CH₂CH-Cl T CH3 naphthalene AlCl3 AlCl3 +Cl-C-C-C-Cl H3C CH3 a,a-dimethylmalonyl dichloride -CH₂Cl AlCl3 (a hydrocarbon C19H16) (a compound with 10 carbons) 1) AlCl3 2) H₂O (three products, all isomers with formula C₁5H12O₂)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (4 reviews)
a Each alkyl halide group reacts with benzene in a separate FriedelCrafts alkylation reaction b The ...View the full answer

Answered By

FREDRICK MUSYOKI
Professional Qualities:
Solution-oriented.
Self-motivated.
Excellent problem-solving and critical thinking skills.
Good organization, time management and prioritization.
Efficient troubleshooting abilities.
Tutoring Qualities:
I appreciate students as individuals.
I am used to tailoring resources for individual needs.
I can integrate IT into student's lessons.
I am good at explaining concepts.
I am able to help students progress.
I have a wide curriculum knowledge.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Give the structures of the principal organic product(s) expected in each of the reactions given in Fig. P16.59 on p. 786, and explain your reasoning. Fig. P16.59 (a) (b) (c) . ACI benzene (large...
-
Give the structures of both the reactive intermediate and the product in each of the following reactions: (a) (b) CH,CH,CCH, + HBr- CH2 CH,CH CCHH rides
-
Give the structures of the products expected when (t) valine and (2) proline (or other compounds indicated) react with each of the following reagents: (a) Ethanol (solvent). H2SO4 catalyst (b)...
-
Compare the ACA passed in 2010 with the Massachusetts health plan adopted in 2006 (use the Internet to obtain more details than we have provided in this chapter). Be sure to note the similarities and...
-
The disposal of equipment for an amount of cash greater than the carrying value of the equipment results in a cash receipt equal to the carrying value of the equipment plus the gain on the disposal,...
-
The trial balance for Prima Consultants Company on December 31, 2010, appears below. The following information is also available: a. Ending inventory of office supplies, $97 b. Prepaid rent expired,...
-
A \(10-\mathrm{kg}\) cart is connected to a \(20-\mathrm{kg}\) cart by a relaxed spring of spring constant \(1000 \mathrm{~N} / \mathrm{m}\), and both carts are placed on a low-friction track. You...
-
Taussig Technologies Corporation (TTC) has been growing at a rate of 20% per year in recent years. This same growth rate is expected to last for another 2 years, then decline to gn = 6%. a. If D0 =...
-
Curtiss Construction Company, Incorporated, entered into a fixed-price contract with Axelrod Associates on July 1, 2024, to construct a four-story office building. At that time, Curtiss estimated...
-
Would 1-methoxynaphthalene nitrate more rapidly or more slowly than naphthalene at (a) Carbon-4; (b) Carbon-5; (c) Carbon-6? Explain your reasoning. 5 OCH3 N 3 1-methoxynaphthalene
-
The solvolysis reaction of 2-bromooctane in ethanol is relatively slow. However, this reaction is accelerated by the addition of silver ion (as AgNO 3 ), and one of the products is AgBr. Explain how...
-
The purpose of this case is to introduce you to the information available on the website of the International Accounting Standards Board (IASB). Required: Access the IASB home page on the Internet....
-
Jackson Jumper has owned a beach house for the last 20 years. In 2019, a hurricane destroyed his beach house and the beach was determined to be a federally declared disaster area. His adjusted basis...
-
What happens to the basis of an asset that has a recapture of an investment credit?
-
In 2019, Sally Morris, a single taxpayer, pays $3,000 of interest on qualified student loans. Her AGI is $40,000. What is her qualified student loan interest deduction in 2019?
-
Are taxpayers allowed a credit on the total expenditure for research?
-
Managers at the Smooth Ride Car Rental Company are interested in the mean number of miles that people drive per day. From past experience, they know that the standard deviation is 75 miles. A random...
-
In manufacturing, does worker productivity drop on Friday? In an effort to determine whether it does, a company's personnel analyst randomly selects from a manufacturing plant five workers who make...
-
How will relating product contribution margin s to the amount of the constrained resource they consume help a company maximize its profits?
-
On the basis of the hybrid orbitals involved in the bonds, arrange the bonds in each of the following sets in order of increasing length. C--C single bond of propan; C--C single bond of propyne; C--C...
-
Rank the anions within each series in order of increasing basicity, lowest first. Explain. CH(CH2) CH CH
-
Using simple observations or chemical tests with readily observable results, show how you would distinguish between the compounds in each of the following pairs. (a) l-hexyne and 2-hexyne (b) propyne...
-
Alter, Inc. had two jobs in process on July 1, Job 851 and 853. The company uses job order costing and applies manufacturing overhead based on machine hours. The company expected 40,000 machine hours...
-
FitchPo company reports in its 2019 10-K, sales of $565 million and cost of goods sold of $226 million. For next year, you project that sales will grow by 6% and that cost of goods sold percentage...
-
Mr. Cord owns four corporations. Combined financial statements are being prepared for these corporations, which have intercompany loans of $200,000 and intercompany profits of $500,000. What amount...

Study smarter with the SolutionInn App


