Would 1-methoxynaphthalene nitrate more rapidly or more slowly than naphthalene at (a) Carbon-4; (b) Carbon-5; (c) Carbon-6?
Question:
Would 1-methoxynaphthalene nitrate more rapidly or more slowly than naphthalene at
(a) Carbon-4;
(b) Carbon-5;
(c) Carbon-6?
Explain your reasoning.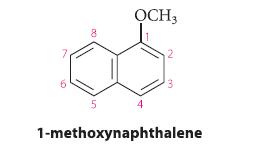
Transcribed Image Text:
5 OCH3 N 3 1-methoxynaphthalene
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
a Because carbon4 is para to the methoxy group the carbocation intermediate involved in nitration ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Would l-methoxynaphthalene nitrate more rapidly or more slowly than naphthalene at carbon-5 Explain your reasoning.
-
1. How strong are the competitive forces confronting J. Crew in the market for specialty retail? Do a [Michael Porter] five-forces analysis to support your answer. (see chapter 3 in the textfor...
-
Carol Harris, Ph.D, CPA, is a single taxpayer and she lives at 674 Yankee Street, Durham, NC 27409. Her Social Security number is 793-52-4335. Carol is an Associate Professor of Accounting at a local...
-
For Table 24.1, suppose you think that you omitted a relevant variable, physicians per capita. Would this harm your results for S or H? Explain your view. Table 24.1 (Model 1) The Effect of Community...
-
Derive sales revenue from data the statement of cash flows and balance sheet Microsoft Corporation reported a balance of $5,196 million in accounts receivable at the beginning of the year and $5,334...
-
Prepare the monthly income statement, monthly statement of owner's equity, and the balance sheet at August 31, 2011, for Alvin Cleaning Company from the data provided in the adjusted trial balance...
-
A 5. 0-kg block suspended from a spring scale is slowly lowered onto a vertical spring (Figure P8.94). (a) What does the scale read before the block touches the vertical spring? (b) If the scale...
-
Assume Polaris invested $ 2.12 million to expand its manufacturing capacity. Assume that these assets have a ten-year life, and that Polaris requires a 10% internal rate of return on these assets....
-
The headteacher at X Primary School finds it very difficult to complete her end-of-term submissions, and this results in confusion and anxiety because of the large volume of outstanding tasks. (1)...
-
A DielsAlder reaction of 2,5-dimethylfuran and maleic anhydride gives a compound A that undergoes acidcatalyzed dehydration to give 3,6-dimethyl phthalic anhydride (see Fig. P16.65). (a) Deduce the...
-
Give the structures of the principal organic product(s) expected in each of the reactions given in Fig. P16.61, and explain your reasoning. (b) (d) benzene (large excess) + ClCH- benzene (large...
-
LeBlanc Inc. shows a balance of $519,289 in the Accounts Receivable account on December 31, 2020. The balance consists of the following: Instalment accounts due in 2021...
-
Model and solve the aggregate planning problem as a linear program.
-
A 90%confidence interval for the population mean time (in minutes) needed to finish a certain assembly process is 90 < x < 130. (a) Sketch this interval, indicating the margin for sampling error....
-
On December Bernie took out a 15-year loan for $300,000. He paid 2% in points in order to get the loan, or $6,000. This is essentially prepaid interest. If the loan is used to purchase his principal...
-
How does the rehabilitation credit differ from the investment credit?
-
How does an investment credit affect the basis of an item purchased?
-
A manufacturer uses two machines to drill holes in pieces of sheet metal used in engine construction. The workers who attach the sheet metal to the engine become inspectors in that they reject sheets...
-
Gordon and Lisa estimate that they will need $1,875,000 in 40 years for their retirement years. If they can earn 8 percent annually on their funds, how much do they need to save annually?
-
Give the principal products expected when 4-octyne or the other compounds indicated are treated with each of the following reagents: (a) H2, Pd/C catalyst (b) H2, Lindlar catalyst (c) Product of (b)...
-
In its latest catalog, Blarneystyne, Inc., a chemical company of dubious reputation specializing in alkynes, has offered some compounds for sale under the following names. Although each name...
-
Build a model of cyclodecyne. Compare its stability qualitatively to that of cyclohexyne; explain your answer.
-
What kind of report provides data or findings, analyses and conclusions, and may also provide recommendations if requested?
-
Inventory is often referred to as the graveyard of business because over - investment in stock is a frequent cause of business failure. Reflecting on what you have learned in this module what is...
-
Jessy bought a mutual fund with a $10 NAVPS and a 2.5% front-end commission. The following year, he sold the fund for $15. What was his return net of fees? Assume no distributions. Round your answer...

Study smarter with the SolutionInn App


