In each of the following pairs, one of the glycols is virtually inert to periodate oxidation. Which
Question:
In each of the following pairs, one of the glycols is virtually inert to periodate oxidation. Which glycol is inert? Explain why.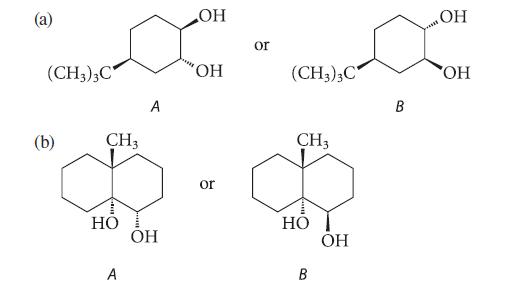
Transcribed Image Text:
В (с (CH3)3C (b) CH3 HO A A OH OH OH or or (CH3)3C CH3 HO B OH B OH OH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
a Because the large tertbutyl group virtually locks the molecule into the chair confo...View the full answer

Answered By

Deborah Joseph
My experience has a tutor has helped me with learning and relearning. You learn everyday actually and there are changes that are made to the curriculum every time so being a tutor has helped in keeping me updated about the present curriculum and all.
I have also been able to help over 100 students achieve better grades particularly in the categories of Math and Biology both in their internal examinations and external examinations.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
In each of the following pairs , one. of the glycols is virtually inert to periodate oxidation. Which glycol is inefi? Explain why. CH3 CH, or
-
Which compound in each of the following pairs would have the higher boiling point? Explain your answers. (a) (b) (c) (d) (e) (f) (g) (h) Hexane, CH3(CH2)4CH3, or nonane, CH3(CH2)7CH3 (i) OH or HO OH...
-
In each of the following pairs of substances, one is stable and known, whereas the other is unstable. For each pair, choose the stable substance, and explain why the other compound is unstable. a....
-
The following question is designed to highlight key concepts from the Loyalty Programs topic article titled, "StarBUCKS, Loyalty, and Breakage" (Nevraumont 2019). Q. Author's position. "If you hire...
-
In an inflationary period, which inventory cost flow method will produce the highest net income? Explain.
-
You are the senior on an audit team for a large public company. This is your second year on the engagement and you have established a good working relationship with some of the clients staff. Your...
-
Regional Highway Transport is a large trucking company. Regional Highway Transport uses the units-of-production (UOP) method to depreciate its trucks. In 2009, Regional Highway Transport acquired a...
-
After five years with a national CPA firm with mostly large manufacturing clients, Amy Kimbell joined Hi-Quality Productions Inc. (Hi-Q) as manager of Manufacturing Accounting. Amy has both CPA and...
-
On March 2 5 , a 6 - month note for $ 1 0 , 0 0 0 was made at 7 % . What was the present value of the note on Jun 1 9 if money is worth 9 % ?
-
Draw a curved-arrow mechanism for each of the conversions shown in Fig. P11.77. Figure P11.77 S
-
(a) Give a curved-arrow mechanism for the reaction shown in Fig. P11.70. Be sure your mechanism indicates the role of the weak acid ammonium chloride. (b) Why does the reaction of an aziridine...
-
Express the following quantities in SI units: (a) 160 acre, (b) 15 gallons (U.S.), (c) 240 miles, (d) 79.1 hp, (e) 60.3 F
-
Give three examples to support the statement "longrange interaction forms the underlying basis for most life processes and technology.
-
Describe the origin and purposes of Title IV-D agencies.
-
It all started with a Facebook post. David Heath was scrolling Facebook one day when he read that socks are the number one item people in homeless shelters request. He remembers thinking that that...
-
Presented below and on page 1004 are two independent situations related to future taxable and deductible amounts resulting from temporary differences existing at December 31, 2014. 1. Mooney Co. has...
-
What is the difference between a firm's gross profit margin, operating profit margin, and net profit margin?
-
Roses to Go is a flower farm that specializes in fragrant roses for florist shops. REQUIRED A. List five joint costs that are likely to be incurred by Roses to Go in raising roses. B. The roses are...
-
A bar of a steel alloy that exhibits the stress-strain behavior shown in Figure 6.22 is subjected to a tensile load; the specimen is 375 mm (14.8 in.) long and has a square cross section 5.5 mm (0.22...
-
Choline, a component of the phospholipids in cell membranes, can be prepared by SN2 reaction of trimethyl amine with ethylene oxide. Show the structure of choline, and propose a mechanism for...
-
Cyclopentamine is an amphetamine-like central nervous system stimulant. Propose a synthesis of Cyclopentamine from materials of five carbons orless. CH3 -CH2CHNHCH3 Cyclopentamine
-
Tetracaine is a substance used medicinally as a spinal anesthetic during lumbar punctures (spinal taps). (a) How would you prepare tetracaine from the corresponding aniline derivative, ArNH2? (b) How...
-
Explain how competition impacts trading in an international market How is public relations different from advertising and sales promotion?
-
Discuss the relationship among quality, value and satisfaction and their roles for developing a long-term relationship with customers. Explain in details.
-
The terms below are just a few terms needed in discussing the world of social media marketing and it is important that students have a clear and consistent understanding of these terms. We will use...

Study smarter with the SolutionInn App


