Predict the major product in each case that would be obtained when the following epoxide reacts with
Question:
Predict the major product in each case that would be obtained when the following epoxide reacts with water under
(a) Basic conditions;
(b) Acidic conditions.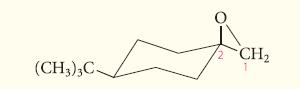
Transcribed Image Text:
(CH3)3C- CH₂
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
As the preceding summary suggests when attempting to predict the products of an epoxide ringopening ...View the full answer

Answered By

Shem Ongek
I am a professional who has the highest levels of self-motivation. Additionally, I am always angled at ensuring that my clients get the best of the quality work possible within the deadline. Additionally, I write high quality business papers, generate quality feedback with more focus being on the accounting analysis. I additionally have helped various students here in the past with their research papers which made them move from the C grade to an A-grade. You can trust me 100% with your work and for sure I will handle your papers as if it were my assignment. That is the kind of professionalism that I swore to operate within. I think when rating the quality of my work, 98% of the students I work for always come back with more work which therefore makes me to be just the right person to handle your paper.
4.80+
174+ Reviews
426+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Predict the major product (or products) that would be obtained when each of the following compounds is nitrated: (a) (b) (c) OH CF CN SO3H OCH3 NO2
-
Predict the major product (or products) formed when each of the following reacts with a mixture of concentrated HNO3 and H2SO4. (a) (b) (c) 4-Chlorobenzoic acid (d) 3-Chlorobenzoic acid (e)...
-
The S N 2 reaction between a Grignard reagent and an epoxide works reasonably well when the epoxide is ethylene oxide. However, when the epoxide is substituted with groups that provide steric...
-
A manufacturing company reports the following information for the month of May. Note: Assume all raw materials were used as direct materials. Activities for May Advertising expense Raw materials...
-
Domitian Corporation accounts for uncollectible accounts receivable using the allowance method. As of December 31, 2011, the credit balance in Allowance for Bad Debts was $170,000. During 2012,...
-
1. For the prototype user interface, design a switchboard that leads from main sections to subsections of related system tasks. The main sections may include member information, registration,...
-
Develop the appropriate primary research question to be associated with this design. Develop a hypothetical research scenario that would necessitate the use of the Action Research Approach and a...
-
This problem is based on the 2008 annual report of Intel Corporation in the appendix. Find in the Selected Financial Data (also known as the Five-Year Financial Summary), or calculate, the following...
-
31 g (20%) A three-mirror ring cavity has mirrors with R = R = 0.8 and R = 0.9 which are separated at 12 = 0.5 m and 123 = 3 = 0.3 m. A glass rod that has a length of 1 = 0.2 m and a refractive index...
-
What is the stereochemistry of the 2,3-butanediol formed when meso-2,3-dimethyloxirane reacts with aqueous sodium hydroxide?
-
Arrange the ions in the following list in order of increasing acidity, and explain your reasoning. | A H3C B CH3 H3C C H D
-
An air compressor takes in air at 14 lbf/in 2, 60 F and delivers it at 140 lbf/in 2, 1080 R to a constant-pressure cooler, which it exits at 560 R. Find the specific compressor work and the specific...
-
Assume that a bank can borrow or lend money at the same interest rate in the LIBOR market. The 90-day rate is 10% per annum, and the 180-day rate is 10.2% per annum, both expressed with continuous...
-
Suppose that, on October 24, 2012, a company sells one April 2013 live cattle futures contract. It closes out its position on January 21, 2013. The futures price (per pound) is 91.20 cents when it...
-
On July 1, 2012, a Japanese company enters into a forward contract to buy \(\$ 1\) million with yen on January 1, 2013. On September 1, 2012, it enters into a forward contract to sell \$1 million on...
-
Suppose that in September 2012 a company takes a long position in a contract on May 2013 crude oil futures. It closes out its position in March 2013. The futures price (per barrel) is \(\$ 68.30\)...
-
Suppose that the term structure of risk-free interest rates is flat in the United States and Australia. The USD interest rate is 7% per annum and the AUD rate is 9% per annum. The current value of...
-
Fernando has been an attorney in the legal department of Mega Manufacturing, Inc., for the past 15 years. As part of Megas restructuring plan, he will lose his job as an employee of Mega, but he will...
-
On October 1, 2014, the Dow Jones Industrial Average (DJIA) opened at 17,042 points. During that day it lost 237 points. On October 2 it lost 4 points. On October 3 it gained 209 points. Deter-mine...
-
Draw the complete structure of the deoxyribonuclcotide sequence from which the mRNA codon in Problem 28.24 was transcribed. Problem 28.24 UAC is a codon for tyrosine it was transcribed from ATG of...
-
Give an mRNA sequence that will code for synthesis of metenkephalin. Tyr-Gly-Gly- Phe-Met
-
Give an mRNA sequence that will code (or the synthesis of angiotensin II. Asp-Arg-Val-Tyr-IIe-His-Pro-Phe
-
In 2014, a failure in a container belonging to the Grupo Mxico mining company caused a spill of 40,000 cubic meters of acids and minerals into the Bacanuchi and Sonora rivers, affecting approximately...
-
Explain which closing disclosure calculations of closing cost details (under loan costs and other costs) are incorrect, show the corrected calculations, and the actual math used to make your...
-
A Maryland public school classroom teacher holding a standard or advanced professional certificate may claim a nonrefundable credit against state income tax for up to what amount of their...

Study smarter with the SolutionInn App


